by Carle Terrasse and Sarah Stern
Introduction
F.tularensis is an intracellular, nonmotile coccobacillus responsible for the zoonotic (transmissible from animal to human) disease tularemia. It was discovered in 1911 as the cause of a fatal, quickly spreading disease in squirrels in California. It can be divided into 3 main subspecies (figure 1), but not all cause disease in humans. In fact mostly subspecies tularensis and holarctica cause human tularemia, while mediasiatic does not. In North America, F. tularensis tularensis is the most common. This bacterium naturally occurs in small mammals, such as rodents and rabbits that mainly acquire it via arthropod bites. As a result, tularemia is primarily observed in rural areas where small rodents and ticks are abundant. As it is easily spread, highly infective, and deadly, F. tularensis is subject of much concern with regards to bioterrorism. In fact, inhalation of only 10 organisms is enough to cause acute respiratory problems and mortality.

Figure 1: Diagram of the evolution of the 3 subspecies holartica, mediasiatica and tularensis (enclosed in light red) of F.tularensis (enclosed in dark red). Adapted from: Birdsell DN, et al. TaqMan Real-Time PCR Assays for Single-Nucleotide Polymorphisms Which Identify Francisella tularensis and Its Subspecies and Subpopulations. PLoS ONE 2014, 9(9):e107964.
Disease
Depending on the subspecies of F. tularensis, the symptoms can be more or less severe. In general, fever, headaches, body aches and malaise develop within the first 3 to 5 days of infection. The different routes of infection lead to more specific symptoms. A human infection can develop from direct contact with rodents, bites from the ticks and mosquitoes, inhalation of aerosolized bacteria and ingestion or contact with contaminated environment but human-to-human transmission has never been seen. These are the common clinical manifestations of the main forms of tularemia:
- Glandular: vector borne transmitted by ticks. Painful swollen lymph nodes result from the infection. An ulcer may develop at the site of infection (figure 2) in which case it is called an ulceroglandular infection.
- Oropharyngeal: ingestion of contaminated food and/or water. A sore throat, vomiting, diarrhea and neck glands swelling are characteristic symptoms.
- Respiratory or pneumonic: acquired via inhalation. It is the most severe form of tularemia.
- Oculoglandular: direct contamination with the eye may lead to this form of tularemia. It is characterized by pain and swelling of the eye. An ulcer may develop too.
- Typhoidal: this form results from an unspecified route of infection and patients will develop a systemic fever where fever, fatigue and chills are the main symptoms.
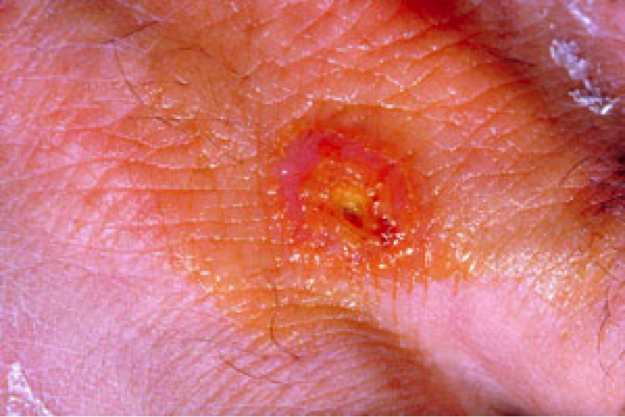
Figure 2: Ulcer formation at the site of infection by F. tularensis. Source: CDC Public Health Image Library (ID #2037).
Regardless of the initial site of contamination, in humans the bacteria will spread to the lymph nodes, liver and spleen after entering macrophages. Macrophages are immune system cells that recognize and engulf target cells for destruction. F. tularensis is able to evade the mechanisms within the macrophages that would normally digest it and effectively uses these cells to taxi around the body.
Epidemiology
Francisella tularensis is naturally present in North America. In the United States, Eastern cottontail rabbits are the main reservoirs of the disease. These rabbits also share a range with Canada, so the disease crosses country lines. Here in Canada, snowshoe hares also carry F. tularensis. There have been recent outbreaks across North America, Asia and Europe (Russia, Kosovo, Germany, Austria, and Italy). These outbreaks are significant enough that tularemia is now classified as a re-emerging disease worldwide. In 2000 and 2003 Kosovo saw large outbreaks of more than 300 cases of oropharyngeal tularemia each year. Recent outbreaks occurred in Turkey and Italy due to contaminated well water. Keep an eye on tularemia; this disease is sure to make the news in the near future!
Virulence Factors
Once F. tularensis gets inside the body, it uses some tools to evade the immune system. The host works hard to destroy the pathogen. One way the body tries to rid itself of the bacteria is by a set of proteins called the complement system. These proteins target the pathogen and aim to lyse it by poking holes in the membrane of the bacteria. F. tularensis has adaptations to deal with this problem. The first is the production of a capsule to avoid detection and binding by complement proteins. If the complement protein C3 does happen to bind, F. tularensis is able to cleave the protein, which renders it inactive.
Another aspect of the host defense that F. tularensis has to deal with is antimicrobial peptides. Antimicrobial peptides are positively charged molecules that kill bacteria. These molecules are attracted to the negative surface of bacteria. F. tularensis modifies one of its surface glycolipids, called Lipid A, to make its surface less negative. This tricks the antimicrobial peptides, and they won’t go after the bacteria.
As soon as 1 hour after infection, F. tularensis invades phagocytic cells called macrophages. Macrophages essentially eat up the bacterium. Inside these cells, the bacterium is safe from extracellular defenses. But how does F. tularensis keep from being digested? After being eaten by a macrophage, F. tularensis is trapped inside a digestive pocket called a phagosome. The phagosome acidifies and gains enzymes and reactive oxygen species to digest the material inside of it. F. tularensis has virulence genes that code for products to combat both the acidity and reactive oxygen species inside the phagosome. To combat phagosome acidification, F. tularensis prevents the phagosome from receiving certain acid hydrolase enzymes, so the pH does not fall. In addition F. tularensis produces the acid phosphatases AcpA, AcpB, AcpC, and Hap that degrade reactive oxygen species.
The last step for F. tularensis is to escape from the phagosome to the cytosol, where it reproduces. The exact mechanism of how this occurs is still unknown. One thought is that the bacteria creates a “needle” and the pokes through the phagosome. Regardless of the method, scientists know that the genes for this process are encoded in the Francisella pathogenicity island (FPI), which codes for a bunch of virulence factors. These virulence factors are key in the pathogen’s stealthy evasion of the immune system and journey to replication.
Treatment
So you have tularemia… quick, get to the doctor! This disease can be fatal, so early detection is important. As F. tularensis has the potential to be used for bioterrorism, your doctor must report any incident to the government. Doctors commonly use antibiotics to treat tularemia. Steptomycin is the drug of choice, followed by tetracyclines such as doxycycline. Surgery may be needed to remove ulcers if the infection is ulceroglandular.
When it comes to tularemia, prevention is key. While a vaccine that uses live attenuated bacteria exists, it is not safe for public use. Because F. tularensis is endemic to North America, take care when hiking or working outdoors. Wear protective clothing, and use insect repellent. Do not handle wild animal carcasses without gloves. Take these precautions and you will be saved from tularemia!
References
CDC: Tularemia[Internet]. Atlanta (GA): Centers for Disease Control and Prevention: c2015 [cited 2015 Nov 15]. Available from: http://www.cdc.gov/tularemia/
D’Alessandro D, Napoli C, Nusca A, Bella A , Funari E. Human tularemia in Italy. Is it a re-emerging disease?. Epidemiology and Infection 2015. 143: 2161-2169. doi:10.1017/S0950268814002799.
Dankova V, Balonova L, Straskova A, Spidlova P, Putzova D, Kijek T, Bozue J, Cote C, Mou S, Worsham P et al: Characterization of Tetratricopeptide Repeat-Like Proteins in Francisella tularensis and Identification of a Novel Locus Required for Virulence. Infection and Immunity 2014, 82(12):5035-5048.
Francisella Tularensis (Tularemia). (2014, February 26). Retrieved November 20, 2015, from http://www.upmchealthsecurity.org/our-work/publications/2014/francisella-tularensis-fact-sheet
Jones CL, Napier BA, Sampson TR, Llewellyn AC, Schroeder MR, Weiss DS: Subversion of Host Recognition and Defense Systems by Francisella spp. Microbiology and Molecular Biology Reviews : MMBR 2012, 76(2):383-404.
Kim J-e, Seo Y, Jeong Y, Hwang MP, Hwang J, Choo J, Hong JW, Jeon JH, Rhie G-e, Choi J: A novel nanoprobe for the sensitive detection of Francisella tularensis. Journal of Hazardous Materials 2015, 298:188-194.
Maggio S, Takeda K, Stark F, Meierovics AI, Yabe I, Cowley SC: Control of Francisella tularensis Intracellular Growth by Pulmonary Epithelial Cells. PLoS ONE 2015, 10(9):e0138565.
Santic M, Al-Khodor S, Abu Kwaik Y: Cell biology and molecular ecology of Francisella tularensis. Cellular Microbiology 2010, 12(2):129-139.
Straskova A, Spidlova P, Mou S, Worsham P, Putzova D, Pavkova I, Stulik J: Francisella tularensis type B Delta dsbA mutant protects against type A strain and induces strong inflammatory cytokine and Th1-like antibody response in vivo. Pathogens and disease 2015, 73(8).
Wobeser G, Campbell GD, Dallaire A, McBurney S: Tularemia, plague, yersiniosis, and Tyzzer’s disease in wild rodents and lagomorphs in Canada: A review. The Canadian Veterinary Journal 2009, 50(12):1251-1256.
