f24c1892308e8af34fd1b5c1fea8055c
Category Archives: Uncategorized
c43ef69619b7d873163b828a1d0544c5
f24c1892308e8af34fd1b5c1fea8055c
c43ef69619b7d873163b828a1d0544c5
f24c1892308e8af34fd1b5c1fea8055c
c43ef69619b7d873163b828a1d0544c5
f24c1892308e8af34fd1b5c1fea8055c
Treponema denticola
By Florencia Darino and Alex Lang
Introduction
Treponema denticola is a long spiral-shaped, anaerobic bacterium commonly found in the mouth. T. denticola are gram-negative cells, meaning they have a thin peptidoglycan layer and an outer lipid membrane. T. denticola is highly specialized to reside in the gum crevice of the oral cavity and is one of the bacterial species responsible for causing periodontal disease.
Disease
Periodontal disease, also known as gum disease, is a severe gum infection that affects the soft tissue surrounding teeth. T. denticola, Porphyromonas gingivalis, and Tannerella forsythia are known as the “red complex”. They are the three most common bacteria present in the biofilm (also known as plaque) associated with chronic periodontitis. Together, they help each other grow and thrive during infection.
The disease begins with the accumulation of plaque around teeth. If oral hygiene is poor, plaque can harden under the gumline and become tartar, which can only be removed by a dentist. With time, tartar can cause gingivitis, a reversible, mild version of periodontal disease. Severe periodontal disease occurs once gum tissue breaks down and pockets begin to develop between teeth and gums, filling up with even more plaque.
The protein-degrading (proteolytic) enzymes secreted by T. denticola break down the host gum proteins. The lysis of gingival cells and subsequent inflammation are the primary causes of periodontitis. Symptoms of periodontitis include pain, redness, swelling, and bleeding of the gums, and a receding gum line. If left untreated, the damage caused by T. denticola can eventually lead to tooth loss and the spread of infection to the jaw bone.
T. denticola has also been shown to promote oncogenesis, where healthy cells become cancer cells via genetic and/or cellular changes. A specific enzyme produced by T. denticola was identified in the majority of tumor samples and was shown to degrade regulatory proteins involved in the control of inflammation and tumor microenvironment.
Figure 1. Cross section of gum and tooth with plaque. (Source: Alex Lang, 2021)
Epidemiology
Periodontal disease is one of the most common infectious diseases around the world. According to the Centers of Disease Control and Prevention, about 47% of all adults over 30 in the U.S.A. have some level of periodontal disease. The susceptibility also increases with age, with 70% of people over 65 having the disease. Other factors, such as smoking, medications, hormone changes, and underlying health conditions can also increase the risk of developing periodontitis.
The mouth contains a wide range of natural microbiota. However, periodontal disease often results from the overgrowth of certain bacterial species that are also present in healthy individuals. In fact, studies have shown that it is possible to predict the development of periodontitis based on the abundance of different species, especially those of the Red Complex such as T. denticola.
Virulence factors
T. denticola has a variety of virulence factors that allow it to infect the host. One of the first steps to infection is attachment, and T. denticola cells are covered in major outer sheath proteins, allowing it to bind to the gum tissue. If someone infected brushes their teeth or uses mouthwash, some T. denticola cells will still remain attached. T. denticola also has a very strong metabolic association with P. gingivalis. This means that both species can work together to break down proteins, share nutrients, and become overall more efficient. Studies have shown that when cultured together, biofilm formation is enhanced. Biofilm is a slimy substance that contains a dense population of bacteria. Bacteria secrete gooey extracellular polymeric substance (EPS), which is made up of sugars, proteins, and DNA. This keeps the bacterial cells together, provides attachment to teeth, and can help protect bacteria against environmental stress and harsh conditions.
Like other gram-negative bacteria, T. denticola can release small spherical vesicles filled with various molecules such as enzymes and toxins which further help invasion. When released into the environment, they can help break down the tight junctions between host cells and allow the bacterial cells to penetrate deeper gum tissues. Once inside the gum tissue, bacteria can use their internalized flagella to swim deeper into the tissue. By being located under the outer sheath, the flagella are concealed from the immune system, avoiding recognition by antibodies. T. denticola can also respond to environmental changes by a process called chemotaxis. Bacteria can move towards different stimuli such as glucose, allowing them to make the most out of their surroundings and outcompete other bacteria that do not have this ability.
The host’s body will try to fight off the infection to the best of its ability, but T. denticola has ways of suppressing immune responses. This is highly advantageous to T. denticola, as it can remain unnoticed for longer periods of time. Some human epithelial cells produce antimicrobial peptides, which are molecules that can inhibit and destroy bacterial cells. T. denticola can prevent these molecules from attaching to its outer membrane and entering the cell. If they do end up entering, T. denticola can pump these molecules out before they cause any damage. T. denticola can also break down different cytokines which are used by the body to alert and activate the immune system. Therefore, it can continue growing and causing damage without being noticed by the immune system.
Figure 2. T. denticola morphology and its cellular components. (Source: Florencia Darino, 2021)
Treatment
The treatment of T. denticola infections is dependent on how far the infection has spread. For acute infections that have not reached under the gums, excessive plaque can be physically removed from teeth via scraping and a course of antibiotics. For infections that have spread below the gum line and around the root of the tooth, the area may be surgically cleaned and a tissue graft may be performed. This involves removing skin from the roof of the mouth and covering the areas affected by gumline recession caused by T. denticola. For extreme cases when the infection has spread to the bone, a bone graft may be performed. A bone graft procedure is performed by surgically removing the infected bone and then transplanting synthetic material or fragments of your own bone. This essentially “rebuilds” the jaw and allows the bones to regrow over the previously infected areas.
Good oral care including frequent brushing and flossing is the most effective preventative measure against periodontitis. Yearly dentist visits can also help prevent the progression of the disease by removing tartar and plaque that cannot be easily accessed with a toothbrush.
REFERENCES
Centers for Disease Control and Prevention. (2013). Periodontal disease. Centers for Disease Control and Prevention. Retrieved November 18, 2021, from https://www.cdc.gov/oralhealth/conditions/periodontal-disease.html.
Dashper, S. G., Seers, C. A., Tan, K. H., & Reynolds, E. C. (2010). Virulence factors of the oral spirochete Treponema denticola. Journal of Dental Research, 90(6), 691–703. https://doi.org/10.1177/0022034510385242
Foschi, F., Izard, J., Sasaki, H., Sambri, V., Prati, C., Müller, R., & Stashenko, P. (2006). Treponema denticola in disseminating endodontic infections. Journal of dental research, 85(8), 761-765.
Inagaki, S., Kimizuka, R., Kokubu, E., Saito, A., & Ishihara, K. (2016). Treponema denticola invasion into human gingival epithelial cells. Microbial Pathogenesis, 94, 104–111. https://doi.org/10.1016/j.micpath.2016.01.010
Ishihara, K. (2010). Virulence factors of Treponema denticola. Periodontology 2000, 54(1), 117–135. https://doi.org/10.1111/j.1600-0757.2009.00345.x
Loesche, W. J., & Grossman, N. S. (2001). Periodontal disease as a specific, albeit chronic, infection: diagnosis and treatment. Clinical microbiology reviews, 14(4), 727-752.
Mayo Foundation for Medical Education and Research. (n.d.). Periodontitis. Mayo Clinic. Retrieved November 18, 2021, from https://www.mayoclinic.org/diseases-conditions/periodontitis/symptoms-causes/syc-20354473.
Nazir M. A. (2017). Prevalence of periodontal disease, its association with systemic diseases and prevention. International journal of health sciences, 11(2), 72–80.
Nieminen, M., Listyarifah, D., Hagström, J. et al. 2017. Treponema denticola chymotrypsin-like proteinase may contribute to orodigestive carcinogenesis through immunomodulation. Br J Cancer 118, 428–434 (2018). https://doi.org/10.1038/bjc.2017.409
Treponema denticola
By Florencia Darino and Alex Lang
Introduction
Treponema denticola is a long spiral-shaped, anaerobic bacterium commonly found in the mouth. T. denticola are Gram-negative cells, meaning they have a thin peptidoglycan layer and an outer lipid membrane. T. denticola is highly specialized to reside in the gum crevice of the oral cavity and is one of the bacterial species responsible for causing periodontal disease.
Disease
Periodontal disease, also known as gum disease, is a severe gum infection that affects the soft tissue surrounding teeth. T. denticola, Porphyromonas gingivalis, and Tannerella forsythia are known as the “red complex”. They are the three most common bacteria present in the biofilm (also known as plaque) associated with chronic periodontitis. Together, they help each other grow and thrive during infection.
The disease begins with the accumulation of plaque around teeth. If oral hygiene is poor, plaque can harden under the gumline and become tartar, which can only be removed by a dentist. With time, tartar can cause gingivitis, a reversible, mild version of periodontal disease. Severe periodontal disease occurs once gum tissue breaks down and pockets begin to develop between teeth and gums, filling up with even more plaque.
The protein-degrading (proteolytic) enzymes secreted by T. denticola break down the host gum proteins. The lysis of gingival cells and subsequent inflammation are the primary causes of periodontitis. Symptoms of periodontitis include pain, redness, swelling, and bleeding of the gums, and a receding gum line. If left untreated, the damage caused by T. denticola can eventually lead to tooth loss and the spread of infection to the jaw bone.
T. denticola has also been shown to promote oncogenesis, where healthy cells become cancer cells via genetic and/or cellular changes. A specific enzyme produced by T. denticola was identified in the majority of tumor samples and was shown to degrade regulatory proteins involved in the control of inflammation and tumor microenvironment.
Figure 1. Cross section of gum and tooth with plaque.
Epidemiology
Periodontal disease is one of the most common infectious diseases around the world. According to the Centers of Disease Control and Prevention, about 47% of all adults over 30 in the U.S.A. have some level of periodontal disease. The susceptibility also increases with age, with 70% of people over 65 having the disease. Other factors, such as smoking, medications, hormone changes, and underlying health conditions can also increase the risk of developing periodontitis.
The mouth contains a wide range of microorganisms. However, periodontal disease often results from the overgrowth of certain bacterial species that are also present in healthy individuals. In fact, studies have shown that it is possible to predict the development of periodontitis based on the abundance of different species, especially those of the Red Complex such as T. denticola.
Virulence factors
T. denticola has a variety of virulence factors that allow it to infect the host. One of the first steps to infection is attachment, and T. denticola cells are covered in major outer sheath proteins, allowing it to bind to the gum tissue. If someone infected brushes their teeth or uses mouthwash, some T. denticola cells will still remain attached. T. denticola also has a very strong metabolic association with P. gingivalis. This means that both species can work together to break down proteins, share nutrients, and become overall more efficient. Studies have shown that when cultured together, biofilm formation is enhanced. Biofilm is a slimy substance that contains a dense population of bacteria. Bacteria secrete gooey extracellular polymeric substance (EPS), which is made up of sugars, proteins, and DNA. This keeps the bacterial cells together, provides attachment to teeth, and can help protect bacteria against environmental stress and harsh conditions.
Like other gram-negative bacteria, T. denticola can release small spherical vesicles filled with various molecules such as enzymes and toxins which further help invasion. When released into the environment, they can help break down the tight junctions between host cells and allow the bacterial cells to penetrate deeper gum tissues. Once inside the gum tissue, bacteria can use their internalized flagella to swim deeper into the tissue. By being located under the outer sheath, the flagella are concealed from the immune system, avoiding recognition by antibodies. T. denticola can also respond to environmental changes by a process called chemotaxis. Bacteria can move towards different stimuli such as glucose, allowing them to make the most out of their surroundings and outcompete other bacteria that do not have this ability.
The host’s body will try to fight off the infection to the best of its ability, but T. denticola has ways of suppressing immune responses. This is highly advantageous to T. denticola, as it can remain unnoticed for longer periods of time. Some human epithelial cells produce antimicrobial peptides, which are molecules that can inhibit and destroy bacterial cells. T. denticola can prevent these molecules from attaching to its outer membrane and entering the cell. If they do end up entering, T. denticola can pump these molecules out before they cause any damage. T. denticola can also break down different cytokines which are used by the body to alert and activate the immune system. Therefore, it can continue growing and causing damage without being noticed by the immune system.
Figure 2. T. denticola morphology and its cellular components.
Treatment
The treatment of T. denticola infections is dependent on how far the infection has spread. For acute infections that have not reached under the gums, excessive plaque can be physically removed from teeth via scraping and a course of antibiotics. For infections that have spread below the gum line and around the root of the tooth, the area may be surgically cleaned and a tissue graft may be performed. This involves removing skin from the roof of the mouth and covering the areas affected by gumline recession caused by T. denticola. For extreme cases when the infection has spread to the bone, a bone graft may be performed. A bone graft procedure is performed by surgically removing the infected bone and then transplanting synthetic material or fragments of your own bone. This essentially “rebuilds” the jaw and allows the bones to regrow over the previously infected areas.
Good oral care including frequent brushing and flossing is the most effective preventative measure against periodontitis. Yearly dentist visits can also help prevent the progression of the disease by removing tartar and plaque that cannot be easily accessed with a toothbrush.
REFERENCES
Centers for Disease Control and Prevention. (2013). Periodontal disease. Centers for Disease Control and Prevention. Retrieved November 18, 2021, from https://www.cdc.gov/oralhealth/conditions/periodontal-disease.html.
Dashper, S. G., Seers, C. A., Tan, K. H., & Reynolds, E. C. (2010). Virulence factors of the oral spirochete Treponema denticola. Journal of Dental Research, 90(6), 691–703. https://doi.org/10.1177/0022034510385242
Foschi, F., Izard, J., Sasaki, H., Sambri, V., Prati, C., Müller, R., & Stashenko, P. (2006). Treponema denticola in disseminating endodontic infections. Journal of dental research, 85(8), 761-765.
Inagaki, S., Kimizuka, R., Kokubu, E., Saito, A., & Ishihara, K. (2016). Treponema denticola invasion into human gingival epithelial cells. Microbial Pathogenesis, 94, 104–111. https://doi.org/10.1016/j.micpath.2016.01.010
Ishihara, K. (2010). Virulence factors of Treponema denticola. Periodontology 2000, 54(1), 117–135. https://doi.org/10.1111/j.1600-0757.2009.00345.x
Loesche, W. J., & Grossman, N. S. (2001). Periodontal disease as a specific, albeit chronic, infection: diagnosis and treatment. Clinical microbiology reviews, 14(4), 727-752.
Mayo Foundation for Medical Education and Research. (n.d.). Periodontitis. Mayo Clinic. Retrieved November 18, 2021, from https://www.mayoclinic.org/diseases-conditions/periodontitis/symptoms-causes/syc-20354473.
Nazir M. A. (2017). Prevalence of periodontal disease, its association with systemic diseases and prevention. International journal of health sciences, 11(2), 72–80.
Nieminen, M., Listyarifah, D., Hagström, J. et al. 2017. Treponema denticola chymotrypsin-like proteinase may contribute to orodigestive carcinogenesis through immunomodulation. Br J Cancer 118, 428–434 (2018). https://doi.org/10.1038/bjc.2017.409
Fusobacterium Necrophorum
By Emeric Gohou and Muhammad Shariq Imran
Introduction
Fusobacterium necrophorum is an important pathogen in warm blooded beings- more so in animals than in humans. It was first discovered as a causative agent of calf diphtheria in 1884. In 1890, it was isolated from liver abscesses in cattle and lastly, in 1891, was coined as a source of human infections i.e., lemierre’s syndrome.
The interesting aspect to F. necrophorum is that it is a part of the human and cattle microbiota in the oral and gastrointestinal tract. In cattle, it serves an important function of digesting feed and fermenting the lactic acid consumed by the animal. But, it only becomes pathogenic in case of damage to protective surfaces in the body, making it a classic opportunistic bacteria.
Disease
In cattle, F. necrophorum causes liver abscesses (collection of pus) and calf diphtheria (infection impairing tissues involving oral tract). Normally, damage to the protective surface of rumen can be induced by the strongly acidic nature of the rumen i.e., acid-induced ruminitis, or by foreign objects like sharp feed particles. This damage offers F. necrophorum an opportunity to invade and colonize the ruminal wall. Once in the wall, the bacteria gains access to blood or induces abscesses in the ruminal wall, both of which allow the bacteria access to the portal circulation- circulation between liver and rumen. From the portal circulation, the bacteria is filtered by the liver, resulting in an infection and abscess formation in the liver. The abscess mainly consists of degenerated liver cells and immune cells that are impaired by the toxins produced by the bacteria.
In calves, diphtheria is caused in a very similar fashion. This condition is more common in young calves than in older cattle. In case of any damage in the oral cavity, the opportunistic F. necrophorum gains access to the protective layer through to the underlying muscle tissue. Here, with the help of the toxins that it produces, the bacteria causes necrosis i.e., death of the tissue. This leads to abscess formation. Infectious material might also be breathed into the lungs and cause abscesses there, which may result in breathing difficulties, and in extreme cases, death.
Lastly, in humans, F. necrophorum causes Lemierre’s syndrome. F. necrophorum is a commensal bacteria in the oral cavity of humans. It is believed that the bacteria becomes pathogenic if the host’s defenses are impaired by other pathogens. In such a case, the throat becomes infected and the infection reaches the internal jugular vein through the lymphatic spread from surrounding vessels. Here, the bacteria causes thrombophlebitis i.e., an inflammation that causes formation of blood clots.
All the aforementioned processes through which F. necrophorum establishes an infection in cattle, calves and humans, are illustrated in figure 1 for better understanding.
Epidemiology
The incidence of liver abscesses averages around 18-32% in cattle. However, specific research on how much of this is caused by F. necrophorum has not been conducted. On the other hand, the concentration of F. necrophorum is higher in grain-fed cattle , along with the higher incidence of liver abscesses. This implies that a grain-fed cow would potentially be at a higher risk of developing liver abscesses due to F. necrophorum infections.
The epidemiological research on calf diphtheria is very similar. Even though the rate of when F. necrophorum is the causative agent is unknown, it has been concluded that diphtheria is more common in grain-fed calves. It can, once more, be implied that a grain-fed calf is more susceptible to diphtheria due to F. necrophorum infections because of a higher concentration of the bacteria.
Research of incidences of Lemierre’s syndrome in humans have exhibited an average of around 1.4 cases per million population. For reasons unknown, the syndrome is found more often in males than in females with a ratio of 2:1. Moreover, it is most common in healthy teenagers and young adults, but some cases of older age groups have also been reported. Unfortunately, since the early 2000’s, the infection numbers have been rising and mortality rates have been reported between 4-18%.
Virulence factors
F. necrophorum has several methods of avoiding the immune system of the host it is invading. It is equipped with endotoxins, hemolysins, haemagglutinin, adhesins and a capsule, but most researchers agree that the bacteria’s most important virulence factor are its leukotoxins. Leukotoxins are toxins secreted by bacteria that are specialised in inhibiting or destroying immune cells. F. necrophorum’s leukotoxins are cytotoxins capable of destroying hepatocytes, immune cells (macrophages, neutrophils) and ruminal epithelial cells. The toxin helps bacterial invasion by preventing macrophages and neutrophils from phagocytosing the bacteria and destroying the liver cells to allow invasion.
F. necrophorum’s leukotoxin has been found to be a soluble toxin with higher specificity for ruminant neutrophils than those of humans or other animals, though it can still infect those other animals. With a size of 336,000 Da, it is considerably larger than leukotoxins from other bacteria such as Staphylococcus aureus at 38,000 Da or actinobacillus actinomycetemcomitans at 114,000 Da. The operon responsible for the transcription of this protein, called lktBAC, consists of 3 genes. The structural gene of the leukotoxin is lktA, the second gene in the operon.
At low concentrations, the toxin can force systemic death of the affected cells, while at higher concentration it directly causes lysis in the cells. It was found to be more effective against neutrophils than against lymphocytes, especially in ruminants and humans. The toxin showed moderate effects against horse neutrophils, but the neutrophils of other animals such as rabbits and pigs were unaffected. The strains of F. necrophorum collected from bovine rumens were approximately 15 times less leukotoxic than those found in hepatic abscesses. The reason for this is unknown, however it does indicate that either not producing leukotoxins in the rumen is an advantage for the bacteria, or that there is some form of leukotoxin suppression occurring in the rumen.
Treatment
The treatment for a F. necrophorum infection will usually depend on the severity of the infection. The treatment will almost alway involve some form of antibiotic, mainly B-lactams like penicillin or cephalosporin. These drugs act by targeting penicillin-binding proteins (PBPs), which are enzymes that are used by the bacteria to build their cell wall. These B-lactams will weaken the integrity of the bacteria’s cell wall, eventually leading to cell lysis. Some necrophorum bacteria have developed B-lactamase enzymes that allow them to counter the activity of penicillin. The relationship between PBP, B-lactams and B-lactamase is illustrated in figure 2. In these cases, doctors will often prescribe antibiotics that specifically target anaerobic bacteria, like metronidazole or clindamycin.
More serious infections may require the use of surgery as well. If left untreated for too long, the bacteria will cause the formation of necrotic tissue which will need to be removed surgically. Abscesses may also form, which will need to be drained by a doctor as well. In the case of Lemierre’s syndrome, the doctor may also prescribe blood thinners like warfarin or clopidogrel to counteract the bacteria haemagglutinin action. Thankfully, less than 5% of people diagnosed with Lemierre’s syndrome succumb to the disease.
The treatment for cattle is similar to that used in humans. They are also given penicillin, as well as Non-Steroidal Anti-Inflammatory Drugs (NSAIDS) to decrease symptoms of fever and inflammation. Surgery is also used to remove necrotic tissue, as well as tracheostomies. A tracheostomy is an operation that inserts a tube into the throat linking it directly to the outside, and is used in cows with severe inspiratory dyspnea. The survival rate is generally high for cattle treated early, as well as a 60% surgical success rate for cows with advanced infections.
References
Arane K, Goldman RD. Fusobacterium infections in children. Can Fam Physician. 2016;62(10):813-814.
Campbell, J. (2021, November 15). Necrotic laryngitis in cattle – respiratory system. Merck Veterinary Manual. Retrieved November 16, 2021, from https://www.merckvetmanual.com/respiratory-system/respiratory-diseases-of-cattle/necrotic-laryngitis-in-cattle.
Cho, H., Uehara, T., & Bernhardt, T. G. (2014). Beta-lactam antibiotics induce a lethal malfunctioning of the bacterial cell wall synthesis machinery. Cell, 159(6), 1300–1311. https://doi.org/10.1016/j.cell.2014.11.017
Jewell, T. (2017, August 22). Lemierre’s syndrome: Symptoms, treatments, and prognosis. Healthline. Retrieved November 16, 2021, from https://www.healthline.com/health/lemierres-syndrome.
Nagaraja, T. G., Narayanan, S. K., Stewart, G. C., & Chengappa, M. M. (2005). Fusobacterium Necrophorum infections in animals: Pathogenesis and pathogenic mechanisms. Anaerobe, 11(4), 239–246. https://doi.org/10.1016/j.anaerobe.2005.01.007
Peek, S. F., Ollivett, T. L., & Divers, T. J. (2018). Respiratory Diseases. In Rebhun’s Diseases of Dairy Cattle (pp. 94–167). Elsevier. https://doi.org/10.1016/B978-0-323-39055-2.00004-8
Riordan, T. (2007). Human infection with Fusobacterium Necrophorum (necrobacillosis), with a focus on Lemierre’s syndrome. Clinical Microbiology Reviews, 20(4), 622–659. https://doi.org/10.1128/cmr.00011-07
Tadepalli, S., Narayanan, S. K., Stewart, G. C., Chengappa, M. M., & Nagaraja, T. G. (2009). Fusobacterium necrophorum: A ruminal bacterium that invades liver to cause abscesses in cattle. Anaerobe, 15(1–2), 36–43. https://doi.org/10.1016/j.anaerobe.2008.05.005Tan’, Z. L., Nagaraja’, T. G., & Chengappa’, M. M. (1996). FUSOBACTERZUM NECROPHORUM INFECTIONS: VIRULENCE FACTORS, PATHOGENIC MECHANISM AND CONTROL MEASURES. 28.
The 2017 Canine Leptospirosis Epidemic in Maricopa County, Arizona 
By: Marco Marcogliese and Maude Tanguay
Introduction:
Canine leptospirosis is categorized as a zoonotic disease which has caused increased concern over the past several decades in a vast majority of countries throughout the world. The disease is mainly caused by spiral, gram-negative bacteria of the genus Leptospira interrogans sensu lato . Such bacteria can be further categorized into antigenically linked serogroups which are sorted into distinct serovars. Serovars are defined as recognizable variations between bacteria or viruses belonging in the same group. As the disease was initially discovered in 1899, it was believed that the serovars icterohaemorrhagiae and canicola were the bacteria mainly responsible for the majority of clinical cases of canine leptospirosis up until 1960.
Therefore, vaccines responsible for targeting the two most popular serovar infections of the disease simultaneously were administered and had seemed to slowly prevent its dissemination across North America. However, over the past two decades, increased incidence of leptospirosis in dogs linked to novel strains has been increasing, with the most common strains reported in the United States included L. kirchneri serovar gripptyphlosa and L. interrogans serovars pomona and bratislava. To this effect, small outbreaks of canine leptospirosis have become more prominent in certain states throughout the United States, more specifically in the Maricopa County of Arizona.
Etiology:
The sudden uptake of reported cases of canine leptospirosis has mainly been attributed to exposure to wild reservoir hosts found in rural or suburban areas. The bacteria are transmitted amongst their hosts via both direct contact with other infected species or indirect contact with substances such as water, soil, food, and infected feces. The main routes of infection involve the penetration of the host’s skin tissue after exposure to contaminated by-products. Once intruding the host’s body, leptospirosis bacteria (i.e. leptospires) invade the bloodstream to infect multiple host sites such as the liver, spleen, kidneys and the central nervous system. These bacteria are highly motile and flexible and are composed of a spiral structure with hook-shaped ends (Figure 1). This infers that they can spread throughout the body rather quickly and attach to various structures within the body and begin replicating.
The amount of damage to such internal organs depends on the servovar’s virulence alongside the host’s vulnerability and amount of exposure. Severe acute infections of the disease can exhibit bacterial colonization of the renal system as their optimal growth conditions are found within the cells that line the renal tubules. This results in critical renal failure due to tubular damage. The bacterium can destroy such cells that are crucial in lining these important intricate tubules found in the kidney. Afterwards, the bacterium is excreted in the urine and further increases the rates of infection as a result.
Figure 1: Electron micrograph of L. interrogans serovar icterohaemorrhagiae
Source: “Centers of Disease Control and Prevention.& R. Weyant (n.d.). Electron micrograph of L. interrogans serovar icterohaemorrhagiae [Photograph]. Public Health Image Library.” https://phil.cdc.gov/details.aspx?pid=1220
Source of Outbreak:
Canine leptospirosis has been a known and common zoonotic disease for over 100 years in America. With cases steadily rising each year in canines, it’s important to understand which factors contribute to the prevalence of outbreaks and its disproportionate rate of incidence in different areas. Understanding the source of outbreaks is also important for public health since canine leptospirosis can be a zoonotic transmitted disease. The environmental factors characterizing different regions are the most noticeable source of outbreaks in multiple studies which have retrospectively studied this disease. In a recent meta-analysis, dogs exposed to large environmental water sources were around 68% more likely to be infected. Stagnant water sources and floods seem to be frequently contaminated with leptospira serovars. The pathogens which are shed through the urine of infected animals survive for long periods of time when found in water, sustaining themselves until they come in contact with a susceptible host’s skin or mucous membranes. Areas inhabited with wildlife such as suburbs, fields or forests have an even higher potential of leptospirosis infection since there are no preventative measures to reduce their rate of transmission. Although studies were able to easily identify different variables contributing to outbreaks, the correlation between the risk of being infected with disease with these variables has yet to be proven.
Cause of Outbreak:
Being a widely known disease and pathogen, canine leptospirosis outbreaks are frequently reported. A notable example of this is the Maricopa County outbreak in Arizona reported in 2017. The state of Arizona has been reporting multiple clusters of the infection in the last few years despite its unfavourable environment. Arizona receives low amounts of annual rainfalls and lacks the humidity encouraging the growth and dissemination of the organisms. Knowing that leptospirosis is less frequent in drier desert environments such as Arizona, public health officials found it crucial to find out the cause of these canine leptospirosis outbreaks.
Veterinarians in Arizona are required to report new cases of leptospirosis. However, a survey showed that only 57% of veterinarians in the state knew when and how to properly report a zoonotic disease. The survey also noticed that veterinarians treating small animals were the least informed on zoonotic disease reporting. Shockingly, leptospirosis is only reported by 42% of the animal health professionals surveyed. Similarly, 12% of the small animal veterinarians of Arizona believed that canine leptospirosis was not a reportable disease.
Socioeconomic factors are also a large reason for outbreaks of the disease in dogs. Studies show that dogs living in lower-income neighborhoods were more exposed to the various environmental factors associated with the disease and had less access to veterinary care and lower vaccination rates. Historically, lower-income areas often have a lesser understanding and knowledge regarding vaccines which makes them hesitant to vaccinate their pets. On the other hand, studies also showed that some middle-class areas had higher infection rates. This was suggested to be caused by an increased amount of testing being possible with the higher income of clients in the area.
The 2017 outbreak presented many cases with novel symptoms less commonly associated with leptospirosis such as conjunctivitis and vomiting. Less than 40% of the cases exhibited signs of fever, the most common clinical sign of canine leptospirosis. This leads to the disease not being tested as quickly as it should be, increasing the chances for an infected animal to contaminate its environment. Essentially, the diagnosis of this disease in canines is quite challenging as it is affected by the stage of the disease, the vaccination status of the individual, the environment and potential exposure to the pathogen. The available tests each have their strengths and weaknesses. Using a combination of potential treatments tends to be the most effective and efficient way to get a quick and accurate result but also more expensive for pet owners.
These studies identified that due to the unfavourable environment in Arizona, veterinarians don’t tend to be as vigilant or up to date with the latest protocols regarding the disease than veterinarians in high risk areas.
Furthermore, this outbreak was composed majoritarily of cases from dogs living in urban areas. This also indicates that different attitudes and practices among veterinarians in different areas most likely heavily impacted the prevalence of the disease.
Preventative Measures:
With veterinarians being the first to work with and diagnose the disease, they need to keep up with the latest strategies and protocols. This is crucial when examining recent outbreaks of diseases such as leptospirosis that occured in Maricopa County in 2017.
The first step to ensure the prevention of the disease in animals as well as humans is increasing disinfection and improving hygiene when there is possible contact with animal urine. These are easy and necessary precautions which should be taken regardless of the area’s risk for leptospirosis. In addition, limiting the amount of contact between dogs and any potentially contaminated water source is also an essential first step in preventing the disease.
The most efficient way for veterinarians to prevent canine leptospirosis is by administering vaccines. Since a multitude of wild animals frequently shed leptospires, it is impossible to assume that domesticated animals could achieve herd immunity. Too many animals acquire the disease from water or soil contaminated by infected wildlife. The vaccine allows for dogs to have antibodies against the disease. These new antibodies allow the immune system to be equipped to quickly clear the infection should the animal come into contact with the pathogen. As seen with the study conducted by Blanchard et al., (Figure 2) introducing annual vaccines which target several of the common serovars associated with leptospirosis can decrease the ability for the organism to colonize the host. This is an effective practice that can reduce the amount of potential reservoirs in an environment as a result. Leptospirosis vaccines should also be mandatory in places such as kennels and groomers to decrease the chances of spreading the disease to more individuals. Infected dogs should be isolated and their urine should be contained and disinfected in order to decrease the chances of infecting another potential host as well.
Finally, veterinarians should be aware of the latest news regarding the disease, especially with the number of cases rising each year. Improving education and awareness about zoonotic diseases will allow veterinarians to prevent, treat and most importantly, manage the incidence rate of canine leptospirosis. Creating a standardized vaccination protocol would be beneficial in reducing the impact of the conflicting approaches between veterinarians. It would also help prevent outbreaks in unsuspecting areas such as Arizona. There is also a need for better communication between veterinarians and public health officials to prevent outbreaks and reduce the number of cases overall.
Figure 2: Comparison of different serovar groups growth associated with bacterial leptospirosis in vaccinated dogs compared to the unvaccinated
Source: “Blanchard, S., Cariou, C., Bouvet, J., Valfort, W., Oberli, F., Villard, S., Barret-Hilaire, F., Poulet, H., Cupillard, L., & Saint-Vis, B. (2021, June 18). [Photograph]. Quantitative Real-Time PCR Assays for the Detection of Pathogenic Leptospira Species in Urine and Blood Samples in Canine Vaccine Clinical Studies: A Rapid Alternative to Classical Culture Methods.” https://journals-asm-org.proxy3.library.mcgill.ca/doi/10.1128/JCM.03006-20
Conclusion:
To conclude, the Maricopa County canine leptospirosis outbreak of 2017 was largely due to veterinarians and health officials not considering the state to be at risk for this disease in dogs due to the dry and desertic climate. However, since wildlife have a large role in spreading the disease, they can cause outbreaks in unsuspecting areas where domesticated animals might be less protected and where prevention against canine leptospirosis isn’t as common. Therefore, as cases rise, it will be important for veterinarians all over to increase the amount of prevention against canine leptospirosis and to increase awareness about the disease in order to reduce the chances for future outbreaks.
References:
André-Fontaine G. (2006). Canine leptospirosis–do we have a problem?. Veterinary
microbiology, 117(1), 19–24. https://doi.org/10.1016/j.vetmic.2006.04.005
Farr R. W. (1995). Leptospirosis. Clinical infectious diseases : an official publication of the
Infectious Diseases Society of America, 21(1), 1–8. https://doi.org/10.1093/clinids/21.1.1
Goldstein, R. E. (2010, November 1). Canine Leptospirosis. ScienceDirect. https://www.sciencedirect.com/science/article/pii/S0195561610000951?via%3Dihub#bib6
Goldstein, R. E., Lin, R. C., Langston, C. E., Scrivani, P. V., Erb, H. N., & Barr, S. C. (2008,
May 1). Influence of Infecting Serogroup on Clinical Features of Leptospirosis in Dogs. Wiley Online Library. https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1939-1676.2006.tb02886.x
Jenni, M. L., Woodward, P., Yaglom, H., Levy, C., Iverson, S. A., Kretschmer, M., Jarrett, N.,
Dooley, E., Narang, J., & Venkat, H. (2019, November 15). Knowledge, attitudes, and practices among veterinarians during an outbreak of canine leptospirosis — Maricopa County, Arizona, 2017. ScienceDirect. https://www.sciencedirect.com/science/article/pii/S0167587719305689
Langston, C. E., & Heuter, K. J. (2003, July 1). Leptospirosis: A re-emerging zoonotic disease.
ScienceDirect. https://www.sciencedirect.com/science/article/pii/S0195561603000263?via%3Dihub
Reagan, K. L., & Skyes, J. E. (2019, July 1). Diagnosis of Canine Leptospirosis. ScienceDirect.
https://www.sciencedirect.com/science/article/pii/S0195561619300385?via%3Dihub
Rentko, V. T., Clark, N., Ross, L. A., & Schelling, S. H. (1992, July 1). Canine Leptospirosis: A
Retrospective Study of 17 Cases. Wiley Online Library. https://onlinelibrary.wiley.com/doi/10.1111/j.1939-1676.1992.tb00345.x
Ricardo, T., Previtali, A., & Marcelo, S. (2020, August 1). Meta-analysis of risk factors for
canine leptospirosis. ScienceDirect. https://www.sciencedirect.com/science/article/pii/S0167587720301513?via%3Dihub
Sessions, J. K., & Greene, C. E. (2004, August). Canine Leptospirosis: Epidemiology,
Pathogenesis, and Diagnosis. Auburn University College of Veterinary Medicine. http://vetfolio-vetstreet.s3.amazonaws.com/mmah/b1/b3b4ac15fb443ebb9d66e46acc513e/filePV_26_08_606_0.pdf
Taylor, C., O’Neill, D. G., Catchpole, B., & Brodbelt, D. C. (2021, May 31). Incidence and
demographic risk factors for leptospirosis in dogs in the UK. British Veterinary Association. https://bvajournals.onlinelibrary.wiley.com/doi/10.1002/vetr.512
Venkat, H., Yaglom, H. D., & Adams, L. (2019, August 1). Knowledge, attitudes, and practices
relevant to zoonotic disease reporting and infection prevention practices among veterinarians â Arizona, 2015. ScienceDirect. https://www.sciencedirect.com/science/article/pii/S0167587718305208?via%3Dihub
Ward, M. P. (2002, December 30). Seasonality of canine leptospirosis in the United States and
Canada and its association with rainfall. ScienceDirect. https://www.sciencedirect.com/science/article/pii/S0167587702001836?via%3Dihub
White, A. M., Zambrana-Torrelio, C., Allen, T., Rostal, M. K., Wright, A. K., Ball, E. C.,
Daszak, P., & Karesh, W. B. (2017, April 1). Hotspots of canine leptospirosis in the United States of America. ScienceDirect. https://www.sciencedirect.com/science/article/pii/S109002331730059X?via%3Dihub
Listeria monocytogenes outbreak in Colorado 2011
Introduction
On September 2, the Centers for Disease Control and Prevention (CDC) was notified of seven cases of listeriosis in Colorado. Nine days later, a total of 15 cases, including one death, were reported from 4 states. All infected persons were hospitalized, and the majority were over 60 years old. The number of cases continued to rise and spread to other states in the following weeks. The multistate investigation revealed that cantaloupes grown at Jensen Farms and marketed as “Rocky Ford” were the source of the outbreak. By the end of the outbreak, there were 147 confirmed cases across 28 states, including 33 deaths and one related miscarriage (Figure 1).
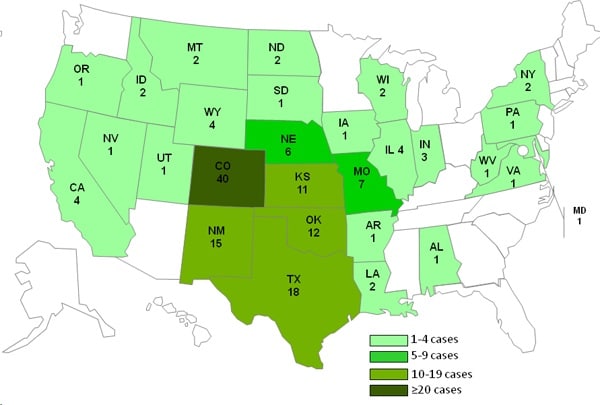
Figure 1: Number of people infected with L. monocytogenes during the 2011 outbreak. Source: Center for Disease Control and Prevention, Case Count Maps (2012).
Listeriosis
Listeriosis is an uncommon but serious infection caused by Listeria monocytogenes. Listeria is ubiquitous in the environment and is transmitted through the consumption of contaminated food products. Outbreaks are most often associated with raw produce, deli meats, and unpasteurized cheese. The elderly, immunocompromised individuals, pregnant women, and newborns are at high risk for an invasive type of infection when the bacterium enters the bloodstream. Neonatal infections may also occur if the bacterium penetrates the placental barrier. Severe symptoms include sepsis, encephalitis, and meningitis. In healthy individuals, listeriosis causes fever and diarrhea that is usually self-limiting.
Source of the outbreak
Whole cantaloupes grown at Jensen Farms in Colorado were identified to be the source of the outbreak. Among patients with information on food consumption, 93% reported eating cantaloupes in the month before the outbreak. The following traceback investigations indicated that the contaminated cantaloupes all came from Jensen Farms and were shipped to at least 24 states between July and September. Moreover, five Listeria subtypes were isolated from Jensen Farms’ packing facility. Subtyping is a powerful method to investigate bacterial transmission by looking at similarities between different isolates of the same bacterial species. In other words, if bacteria isolated from different samples are of the same subtype, it is more likely that they come from a common source. Indeed, the five subtypes from Jensen Farms corresponded to the isolates from the infected persons, the leftover cantaloupes in their homes, and the retail locations where they purchased the melons. Surprisingly, no outbreak-associated subtypes were found in the soil samples collected from where the cantaloupes were grown. The suspected sources of the outbreak were a truck that routinely travels between the processing facility and a nearby cattle farm, low-level contamination of incoming cantaloupes, and persisting bacteria on the processing equipment.
Since the outbreak was linked to whole cantaloupes rather than pre-cut products, there must be substantial numbers of L. monocytogenes that survive and persist on the rind of the fruit. Indeed, follow-up research indicates that the outbreak-associated strains are able to survive and grow on the rind, which somehow supports the growth of Listeria better than the flesh or extracts. This could be explained by the intricate netting surface that promotes bacterial adherence and proliferation. For the pathogens to enter human bodies, they may be introduced to the inside of the fruit where they multiply quickly due to sufficient nutrients and optimum pH levels (6.1-7.1).
Cause of the outbreak
Although the exact cause of contamination remains unknown, the FDA investigation report reveals several problems that might have led to the outbreak (Figure 2). In particular, post-harvest processing and sanitation practices in the packing facility were of major concern. To start with, the cooling and drainage system was poorly designed, which allowed water to collect near the processing area and the employee walkways. The facility floor and packing equipment were also difficult to clean, which gave Listeria a chance to persist in biofilms. Additionally, Jensen Farms upgraded their hydro cooler about a year before the outbreak, as recommended by the auditor. However, the old equipment was replaced by a second-handed machine for processing potatoes, not cantaloupes. Not only was the equipment not suitable for decontaminating melons, but the washing solution also contained no antimicrobial compound of any kind. Finally, the cantaloupes were not pre-cooled to optimal temperature before cold storage. Consequently, energy stored in the fruits may raise the temperature of the storage unit, thus promoting Listeria growth on the rind.
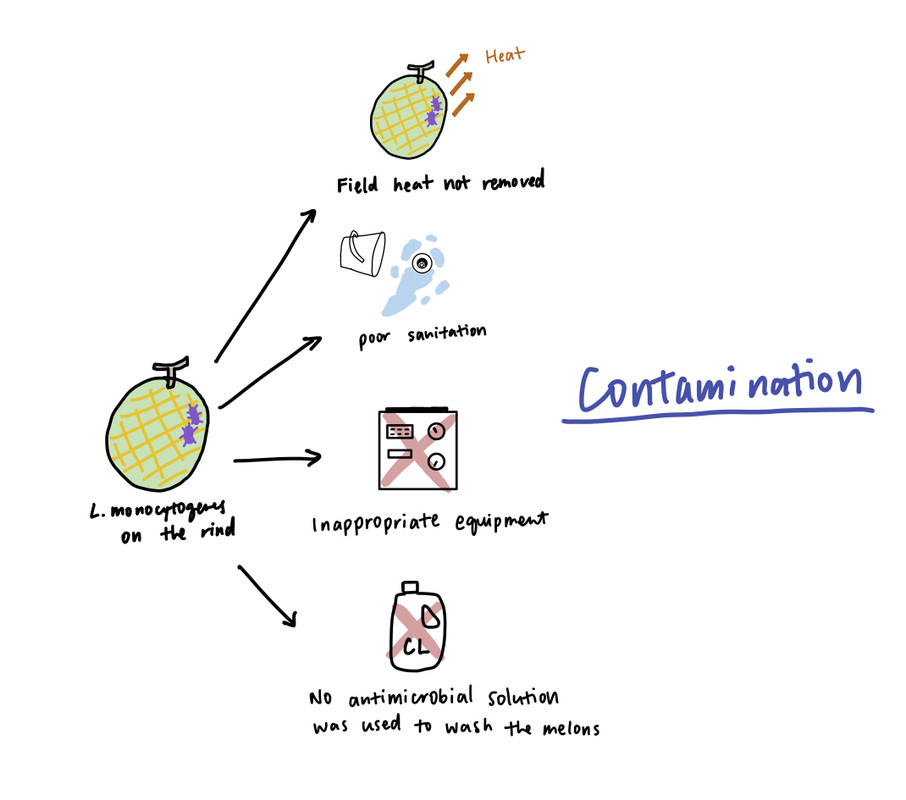
Figure 2: Possible causes of the outbreak. Source: Yuan Zhuang (2021).
Measurement taken to end the outbreak
The public health authorities responded immediately as soon as the first few cases had been reported. The FDA worked closely with the CDC, the firms involved, and public health authorities in states where illnesses occurred to determine the exact source of contamination. Initial interviews of infected persons suggested that the outbreak was most likely associated with eating “Rocky Ford” cantaloupes. This hypothesis was tested and confirmed by laboratory analysis of samples taken at different locations. In response, Jensen Farms issued a voluntary recall of all their whole cantaloupes. The processing facility was also temporarily shut down to prevent additional food contamination. Furthermore, several companies in neighbouring states initiated recalls of products containing cantaloupes produced by Jensen Farms. Meanwhile, the CDC issued a warming that people at high risk for listeriosis, including older adults, persons with weakened immune systems, and pregnant women, should avoid eating cantaloupes from the Jensen Farms.
Aftermath
According to the FDA, the outbreak could have been prevented if Jensen Farms had adhered to FDA guidelines during operation. However, the agency also recognized that the complex procedures in the Food Safety Modernization Act (FSMA), which was signed into law in January 2011, could be difficult to implement in real-life practice. Therefore, after the outbreak, the FDA proposed two rules that address problems found at Jensen Farms. The first rule aims to prevent microbial contamination through better sanitation controls. The second rule emphasizes the development of a hazard-reduction plan that includes four aspects: identification of potential food contamination, actions needed to prevent them, monitoring procedures, and steps required to correct the problem.
References
Centers for Disease Control and Prevention. (2011, November 2). Clinical Features/Signs and Symptoms. Retrieved November 15, 2021, from https://www.cdc.gov/listeria/outbreaks/cantaloupes-jensen-farms/signs-symptoms.html
Centers for Disease Control and Prevention. (2012, August 27). Multistate Outbreak of Listeriosis Linked to Whole Cantaloupes from Jensen Farms, Colorado. Retrieved November 15, 2021, from https://www.cdc.gov/listeria/outbreaks/cantaloupes-jensen-farms/index.html
House Committee on Energy and Commerce. (2012, January 10). Report on the Investigation of the Outbreak of Listeria monocytogenes in Cantaloupe at Jense Farms. https://www.foodsafetynews.com/files/2014/07/Energy-and-Commerce-Committee-Report.pdf
Johnston, L. M., Jaykus, L. A., Moll, D., Martinez, M. C., Anciso, J., Mora, B., & Moe, C. L. (2005). A field study of the microbiological quality of fresh produce. Journal of food protection, 68(9), 1840–1847. doi: 10.4315/0362-028x-68.9.1840
Martinez, M. R., Osborne, J., Jayeola, V. O., Katic, V., & Kathariou, S. (2016). Capacity of Listeria monocytogenes Strains from the 2011 Cantaloupe Outbreak To Adhere, Survive, and Grow on Cantaloupe. Journal of food protection, 79(5), 757–763. doi: 10.4315/0362-028X.JFP-15-498
McCollum, J. T., Cronquist, A. B., Silk, B. J., Jackson, K. A., O’Connor, K. A., Cosgrove, S., Gossack, J. P., Parachini, S. S., Jain, N. S., Ettestad, P., Ibraheem, M., Cantu, V., Joshi, M., DuVernoy, T., Fogg, N. W., Gorny, J. R., Mogen, K. M., Spires, C., Teitell, P., Joseph, L. A., Tarr, C. L., Imanishi, M., Neil, K. P., Tauxe, R. V., & Mahon, B. E. (2013). Multistate Outbreak of Listeriosis Associated with Cantaloupe. New England Journal of Medicine, 369(10), 944-953. doi:10.1056/NEJMoa1215837
Sandora, T. J., Gerner-Smidt, P., & McAdam, A. J. (2014). What’s your subtype? The epidemiologic utility of bacterial whole-genome sequencing. Clinical chemistry, 60(4), 586-588. doi:10.1373/clinchem.2013.217141
Schlech, W. F., III, & Acheson, D. (2000). Foodborne Listeriosis. Clinical Infectious Diseases, 31(3), 770–775. doi: 10.1086/314008
Dietary Zinc Improves our Resistance to Bacterial Infection
By Alessandra Granata and Arianna Cancian-Kavoliunas
Introduction
Typically, when we think of ways our bodies protect us from illness, we might think of things like the cells of our immune system, or our body’s mucus and skin barriers. However, something often overlooked is how proper nutrition also plays a vital role in the body’s ability to ward off pathogens.
Roughly one third of the world population is deficient in dietary Zinc (figure 1), and areas of the world with Zinc deficient populations often experience higher frequencies of bacterial infection. A team of researchers at the University of Adelaide set out to determine if dietary zinc levels affect susceptibility to bacterial infection. Narrowing their search, they specifically explored if there was a correlation between dietary zinc deficiency and susceptibility to infection by Streptococcus pneumoniae, a bacterium that causes a variety of conditions in humans, including pneumonia. Their research confirmed that adequate dietary zinc levels improves the body’s resistance to Streptococcus pneumoniae in humans (figure 2), but how zinc does this is not yet understood.
Figure 1: Global rate of zinc deficiency and mortalities due to zinc deficiency. Source: Arianna Cancian-Kavoliunas
Figure 2: Dietary source of zinc improves the body’s resistance to S. pneumoniae infection. Source: Arianna Cancian-Kavoliunas
How does our body usually fight infections?
Before we can understand how zinc improves our resistance to S. pneumoniae, we need to first understand how we are infected by this bacterium (figure 3) and then how we would usually heal from it (figure 4). S. pneumoniae enters the body through the airways and begins to colonize the surface of tracheal cells, meaning that the bacteria multiply and form colonies on the cells that line the trachea. The infection can then spread to your lower respiratory tract, where some bacterial cells become martyrs. These cells will commit “suicide” in order to release pneumolysins, toxins that destroy host cells including lung epithelial cells. As well, because the body’s immune system has been triggered, a variety of responses are happening. There is an increase in inflammation and blood vessels have become more permeable. S. pneumoniae uses your body’s natural immune response, along with the pneumolysins released by some of its martyrs, to enter your blood. S. pneumoniae can then travel to your brain, causing infection in the form of meningitis via the same mechanism it used in the lungs. As for how the body fights the infection, after inoculation of the host, resident phagocytic cells are recruited to the site of infection. Phagocytic cells are cells that do phagocytosis, which means they use their plasma membrane to engulf a foreign substance. After they are recruited, the phagocytic cells start to phagocytose some of the S. pneumoniae bacteria while also triggering inflammation of the host tissue and emitting an alarm signal. This alarm signal from the phagocytic cells will activate special cells, called T cells, which can then trigger other immune cells, called B-cells, to produce antibodies. Antibodies are Y-shaped proteins that can neutralize pathogens by specifically recognizing a molecule, called an antigen, secreted by the pathogen. The antibodies are released by the B cells and surround the pathogen, which “tags” the Streptococcus pneumoniae bacteria so that our phagocytic cells can recognize and neutralize them more quickly.
Figure 3: Inoculation of host with S.pneumoniae. This image explains how you are infected with the bacteria S. pneumoniae. First, an infected individual spreads S. pneumoniae into the air, and you breathe it in. The bacteria then colonizes the surface of your tracheal cells, and eventually, it is able to spread, through the blood, to other parts of the body. The symptoms commonly associated with severe infection by S. pneumoniae include pneumonia in the lungs, bacteremia as the bacteria grows and travels in the blood, and meningitis in the brain. Source: Arianna Cancian-Kavoliunas
Figure 4: The immune response when S. pneumoniae is found by the immune system. Resident phagocytic cells begin to sense host cell damage, caused by the toxins secreted by S. pneumoniae, using receptors on their cell membrane. Cell damage also allows S. pneumoniae to infiltrate the tissue and be sensed by other receptors on phagocytic cells. The phagocytic cells will then engulf the bacterium, or the segment of the bacterium. As well, the macrophages secrete molecules that signal other cells to start an immune response. These signals trigger an increase in inflammation, cause blood vessels to become more permeable, and even recruit other immune cells to the site of infection. If Zinc is present, it will migrate from the blood into the tissues and potentially induce Zinc toxicity in S. pneumoniae. The macrophages will present the processed segment of S. pneumoniae to naïve T cells. These T cells will then be triggered to form activated T helper cells. These activated T cells can then secrete molecules and activate B cells, which will form antibodies against S. pneumoniae. The antibodies will tag the bacterium for it to be easily phagocytosed. The image displayed represents only one part of the immune response, the body will work tirelessly to try to eliminate the bacterium. Source: Arianna Cancian-Kavoliunas
Why are metal ions important for bacteria?
Metal ions, like Zinc, are necessary for bacteria to grow and develop, but just like most things in life, too much or too little can often be dangerous. S. pneumoniae can often become intoxicated by high levels of Zinc. However, to try to starve the bacterium of Zinc, our bodies have developed really good ways of hiding it. And even so, some bacteria still outsmart us! In an evolutionary arm’s race, bacteria like S. pneumoniae have found ways to steal those ions from us using proteins called metallophores.
What the study showed
The team of researchers from the University of Adelaide, Eijkelkamp et al., were able to show that Zinc significantly affects the number of bacteria and the level of infection present in the individual. The study found that although the number of S. pneumoniae increased 24-36 hours after the infection in both the mice with Zinc deficiency and the mice without, the increase was way more significant in the mice with low levels of Zinc (figure 5). Eijkelkamp et al. also found that 36 hours after infection, there was a lot of Zinc present in the lungs of the mice that had enough dietary Zinc circulating in their blood. They had discovered that Zinc was able to travel from the blood into lung tissue during the infection causing Zinc intoxication to the bacterium only in that specific environment. Zinc intoxication disturbed the level of manganese in S. pneumoniae, which made it harder for the bacterium to thrive. Manganese is an essential metal ion for the growth and survival of S. pneumoniae. This ion normally protects S. pneumoniae from toxic oxidative stress caused by Reactive Oxygen Species (ROS). They also found that Zinc acted as an antimicrobial and helped in the activation of the immune response. Eijkelkamp et al. also found that Zinc was transported into tissues by phagocytic cells, and that the presence of this metal ion had a positive role in increasing inflammation and killing bacterial pathogens. Zinc deficiency is known to decrease the immune response by decreasing the number of functional immune cells like derivatives of T cells. Because the immune system is compromised, the individual has a more difficult time fighting the infection caused by S. pneumoniae.
Figure 5: The experiment and discoveries done by Eijkelkamp et al., 2019: Mice with sufficient zinc levels (left) and mice with low zinc levels (right) were both infected with S. pneumoniae, and the mice with adequate zinc levels had lower tissue damage and lower overall infection spread than the mice with low zinc levels. The susceptibility of the mice to infection by S. pneumoniae was dependent on the level of dietary zinc. Source: Arianna Cancian-Kavoliunas
Why are the study’s findings important?
This study highlights the importance of ensuring that populations in developing countries have access to proper nutrition including a good intake of essential vitamins and minerals. According to the Government of Canada, approximately 2 billion people are not receiving the required amount of vitamins and minerals in their diets! As malnutrition continues to be a growing global concern, other problems related to poor diets have surfaced. Diets that are low in micronutrients, such as zinc, have been shown to increase the risk of acquiring various infections. Another study also found a direct correlation between death from these infections and the level of malnutrition.
Is anyone doing anything to change global zinc deficiency rates?
The WHO funds a World Food Program to ensure that the basic human right, the right to proper nutrition, is met around the world. Programs like this are vital to the prevention and reduction of death and cases of bacterial infection. Zinc supplementation could also be a viable option for mass disease prevention, however the efficacy of supplementation strategies varies, and the reason for this variation is unknown because we still don’t know the exact mechanism zinc uses to prevent bacterial infection. Therefore, more research on the antibacterial mechanism used by zinc would allow for the development and implementation of strategies for the prevention of bacterial infections.
REFERENCES
Eijkelkamp, B. A., Morey, J. R., Ween, M. P., Ong, C.-l. Y., McEwan, A. G., Paton, J. C., & McDevitt, C. A. (2014). Extracellular Zinc Competitively Inhibits Manganese Uptake and Compromises Oxidative Stress Management in Streptococcus pneumoniae. PLOS ONE, 9(2), e89427. doi:10.1371/journal.pone.0089427
Eijkelkamp, B. A., Morey, J. R., Neville, S. L., Tan, A., Pederick, V. G., Cole, N., … & McDevitt, C. A. (2019). Dietary zinc and the control of Streptococcus pneumoniae infection. PLoS pathogens, 15(8), e1007957.
Government of Canada. (2017). Nutrition in developing countries. https://www.international.gc.ca/world-monde/issues_development-enjeux_developpement/global_health-sante_mondiale/nutrition.aspx?lang=eng
Martin, J. E., Lisher, J. P., Winkler, M. E., & Giedroc, D. P. (2017). Perturbation of manganese metabolism disrupts cell division in Streptococcus pneumoniae. Molecular microbiology, 104(2), 334–348. https://doi.org/10.1111/mmi.13630
McGuire S. (2013). WHO, World Food Programme, and International Fund for Agricultural Development. 2012. The State of Food Insecurity in the World 2012. Economic growth is necessary but not sufficient to accelerate reduction of hunger and malnutrition. Rome, FAO. Advances in nutrition (Bethesda, Md.), 4(1), 126–127. https://doi.org/10.3945/an.112.003343
Müller, O., & Krawinkel, M. (2005). Malnutrition and health in developing countries. CMAJ : Canadian Medical Association journal = journal de l’Association medicale canadienne, 173(3), 279–286. https://doi.org/10.1503/cmaj.050342
Prasad A. S. (2008). Zinc in human health: effect of zinc on immune cells. Molecular medicine (Cambridge, Mass.), 14(5-6), 353–357. https://doi.org/10.2119/2008-00033.PrasadWessels, I., Maywald, M., & Rink, L. (2017). Zinc as a Gatekeeper of Immune Function. Nutrients, 9(12), 1286. https://doi.org/10.3390/nu9121286
