by Erica Grier and Mariepièr Glaude
Introduction
Chlamydia trachomatis is a bacterium that infects the columnar epithelial cells of the urethra, cervix and rectum. It also occasionally infects other parts of the human body such as the lungs and eyes, though this is less common. C. trachomatis is gram negative, non-motile and an obligate intracellular pathogen. Infection by C. trachomatis is the most frequently reported STI in Canada and the United States.
Disease
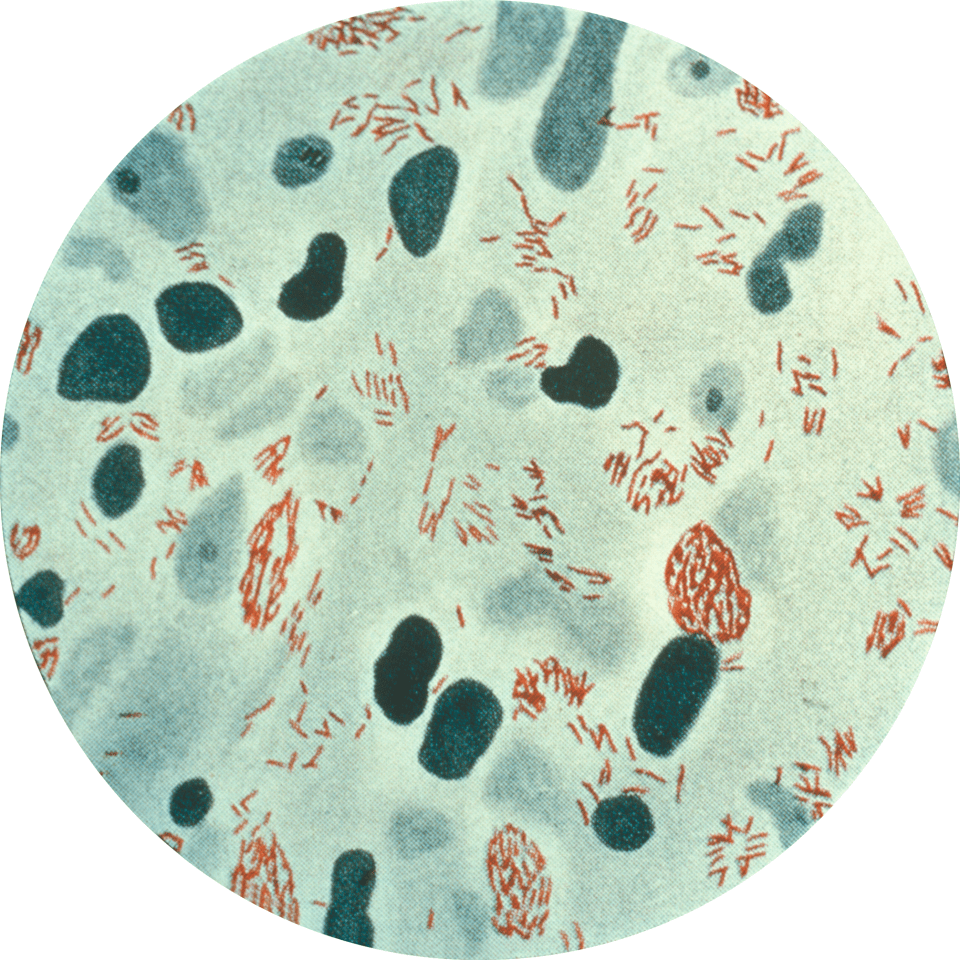
C. trachomatis is spread through vaginal, anal and oral sex. Pregnant women infected with chlamydia can also infect their child while giving birth. Once C. trachomatis gets inside the human host, immune cells will attempt to fight off the pathogen. However, C. trachomatis is able to avoid being killed by immune cells by living and replicating inside of them (Figure 1). Infection by C. trachomatis can cause pain during sex, pain during urination, unusual or bloody vaginal discharge, and sometimes even nausea and mild fevers. However, infection by C. trachomatis can also be completely asymptomatic. Asymptomatic carriers are an important reservoir for the bacteria as people who are unaware they are infected can easily spread the infection to their partners.

Figure 1: Human pap smear showing cells infected with Chlamydia trachomatis at 500x magnification, stained with haematoxylin and eosin (Source: PLOS Pathogens).
Epidemiology
C. trachomatis is found worldwide and is considered endemic to over 50 different countries. Although many C. trachomatis infections occur annually in North America, infections are most common in Africa, the Middle East, India and Southeast Asia.
The most influential factors of infection are sex and age, but race can also play a role in risk of infection. It has been shown that women are more likely to develop chlamydia infections than men. Additionally, the Centre for Disease and Control has recently reported enormous increases in infection rates for both men and women, 37.6% and 29.3% respectively. In a group of 100 000 females ranging from 15 to 19 years old, it is likely that around 2800 of them will be infected by C. trachomatis. For men, the most infected age group is usually older, ranging from 20 to 25 years old. Men who have sex with other men are also at greater risk of contracting C. trachomatis infections. When analyzing risk of infection in terms of race, the rate of C. trachomatis infection is approximately 8 times greater among African American populations than it is among Caucasian populations.
Virulence Factors
For a chlamydia infection to be established, the intracellular pathogen must bind to and be taken in by the host cell. Binding and entry into the host is largely mediated by the polymorphic membrane D (PmpD). This protein acts as an adhesion molecule, allowing the bacteria to bind to and be ingested by the immune cell. Normally, these immune cells kill the pathogen with degradative enzymes upon fusion of the phagosome and lysosome. However, this is not the case when an immune cell is infected with C. trachomatis. C. trachomatis is only able to establish an infection because it is capable of preventing killing by the immune cell.
In order to survive inside the host, C. trachomatis is endocytosed by the immune cell as an elementary body. This body is the explanation behind the intracellular survival of the bacteria. It inhibits fusion of the phagosome to the lysosome, allowing the pathogen to continue its life cycle (Figure 2). The elementary bodies, considered the inactive form of chlamydia, then develop into reticulate bodies. About 20 hours after initial infection, these reticulate bodies divide and differentiate back into elementary bodies that are released and can now induce new rounds of infection.

Figure 2: Elementary bodies inhibit the fusion of the lysosome to the phagosome, preventing the completion of phagocytosis and therefore the degradation of C. trachomatis (Source: Mariepièr Glaude).
Another factor that contributes to virulence is the concept of antigenic variation. Currently, there are 15 known serotypes related to C. trachomatis infections. Antibodies produced for one serotype will have no effect on another serotype, allowing C. trachomatis to remain harmful to host cells. Studies have shown that the lipopolysaccharides (LPS) found in the cell wall of C. trachomatis also play a role in the bacteria’s infectivity. This is because LPS is important in the binding of the bacteria to cells of the genital and respiratory tracts.
Treatment
Chlamydia trachomatis infections can be treated with antibiotics. Azithromycin, erythromycin and doxycycline are commonly used and very effective. These antibiotics clear infections by preventing bacteria from producing proteins essential to survival. Other antibiotics such as amoxicillin are used in place of doxycycline to treat chlamydia in pregnant women because doxycycline can cause harm to a developing child. Patients with chlamydia should abstain from sexual intercourse for seven days after starting antibiotics to avoid infecting their partners. If both partners have C. trachomatis infections they should be treated at the same time to prevent reinfection.
References
Centers for Disease Control and Prevention STD Surveillance [Internet]. 2009. San Francisco (CA): CDC; [updated 2010 Nov 22, cited 2015 Nov 20]. Available from: http://www.cdc.gov/std/stats09/
Darville C. 2015. Chlamydia trachomatis – Urgent need for an effective T cell vaccine to combat the silent epidemic of a stealth bacterial pathogen. PLOS Pathogens [Internet]. [updated 2010 Feb 25, cited 2015 Nov 20]. Available from: http://blogs.plos.org/speakingofmedicine/2015/02/25/ chlamydia-trachomatis-urgent-need-effective-t-cell-vaccine-combat-silent-epidemic-stealth-bacterial-pathogen/
Fadel S, Eley A. 2008. Is lipopolysaccharide a factor in infectivity of chlamydia trachomatis? Journal of Medical Microbiology. 57: 261-266.
Hafner L, Beagley B, Timms P. 2008. Chlamydia trachomatis infection: host immune responses and potential vaccines. Mucosal Immunology. 1(2):116-130.
Miller KE. 2006. Diagnosis and Treatment of Chlamydia trachomatis Infection. American Family Physician. 73(8): 1411-1416.
Mishori R, McClaskey E, Winklerprins J. 2012. Chlamydia Trachomatis Infections: Screening, Diagnosis, and Management. American Family Physician. 86(12): 1127-1132.
Peeling R, Mabey D, Herring A, Hook E. 2006. Why do we need quality-assured diagnostic tests for sexually transmitted infections? Nature Reviews Microbiology. 4: 909-921.
Stamm WE. 1999. Chlamydia trachomatis Infections: Progress and Problems. 179(2): 80-83.
Taylor B, Darville T, Tan C, Bavoil P, Ness R, Haggerty C. 2011. The Role of Chlamydia trachomatis Polymorphic Membrane Proteins in Inflammation and Sequelae among Women with Pelvic Inflammatory Disease. Infectious Diseases in Obstetrics and Genycology. Doi: 10.1155/2011/989762.