by Gaëlle-Laurie Dubréus and Emilie Yeh
Introduction
Mycobacterium leprae, a gram positive bacterium, exists as an obligate intracellular pathogen that causes Hansen’s disease, commonly known as leprosy. This disease has been identified as long as 1550 B.C. in Egypt and was first isolated in 1873 by G.A. Hansen (hence the name of the disease). Since then, much has been discovered on its epidemiology, pathogenicity, and treatments –which were successfully created in the 1970s.
Disease
The mode of transmission of M. leprae appears to be from person-to-person either by droplets from sneezing and coughing or by other nasal secretions. Also, the bacteria can use an abrasion in the skin to colonize the superficial site of the epithelium and the peripheral nerves. Families with members who have leprosy have a higher susceptibility of developing this illness due to genetic predisposition. The only organisms known to harvest the infection and develop the disease are humans and armadillos, whom can remain asymptomatic for upto twenty years (long incubation time).
Although patients usually show signs of sores on the skin, leprosy exists in the following two forms: tuberculoid (pauci-auxillary leprosy), which occurs as skin discoloration, and lepromatous (multi-bacillary leprosy), which manifests as skin lesions, plaques, nodules, thickened skin, and nasal complications. The cause for the manifestation of each is based on the T-cell count, where those with a higher count develop the milder form of leprosy. Lepromatous leprosy is the more severe clinical case so it triggers the production of more antibodies against the bacterium, but with a compromised immune cellular response. Moreover, both forms of leprosy can result in collateral damage to the peripheral nervous system. This can permanently affect the arms, legs, eyes, nerves, and the skin.
Epidemiology
Leprosy is present worldwide, especially in warm tropical and subtropical regions. In 2013, based on statistical analyses from five areas of the World Health Organization (WHO), which included 103 countries, there were about 180.6 thousand cases of leprosy reported. Within a community, the emergence of new cases depends on the rate of transmission between the individuals of that society. For instance, this disease seems to be predominant in certain endemic regions; 14 different but specific countries, such as India, Brazil, several areas in Africa, etc., appear to be contributing to approximately 96% of the new cases of leprosy yearly.
Although adults are less likely to acquire the infection, those who do develop the disease have the lepromatous form, which occurs more frequently in men in comparison to women. Conversely, the tuberculoid form of leprosy evolves more quickly in children and its development is equal among both sexes. Since treatment with multi-drug therapies (MDT) debuted, the prevalence of this disease has significantly diminished as in less than 1 per 10,000 individuals. However, in developing countries, there exists a lack of education on this disease as well as low-to-no access to treatment. Consequently, those carrying the bacterium may not be aware of their infection and suffer from severe physical complications and handicaps in the long run– due to late diagnosis.
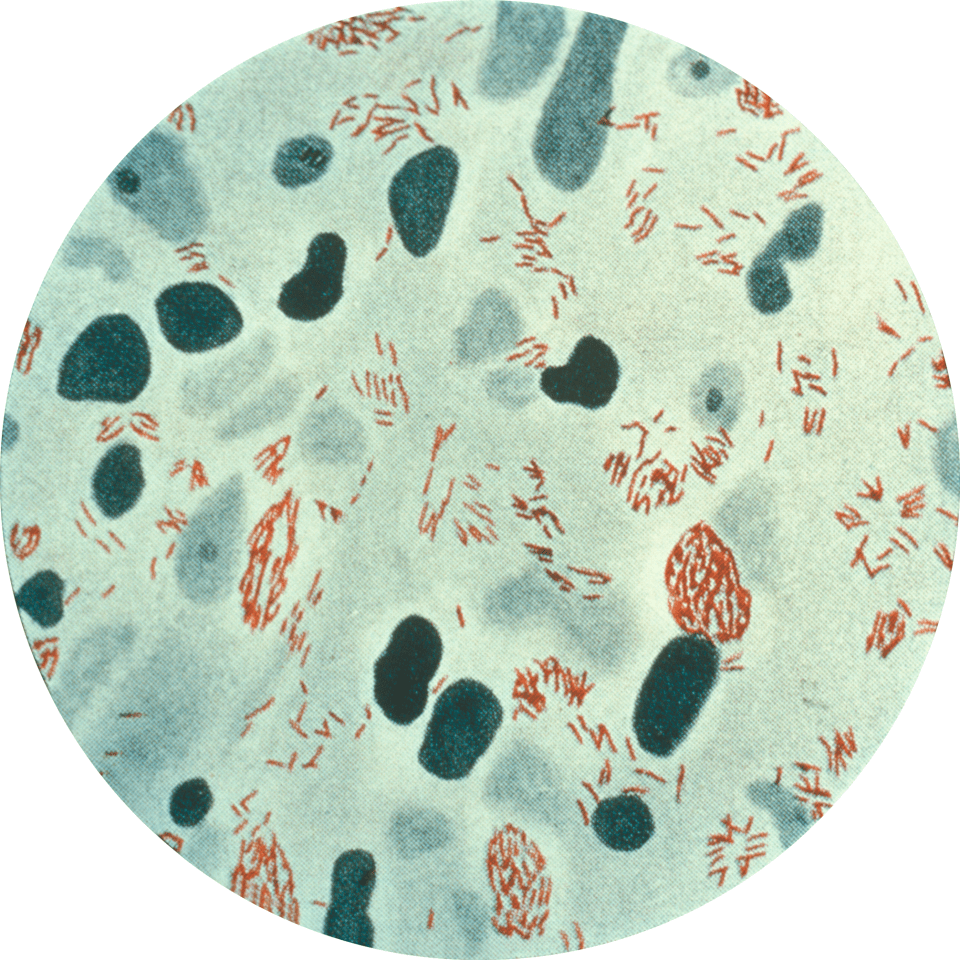
Figure 1: A biopsy from the skin lesion of a patient infected with M. leprae. Here, the bacteria are stained red. (Source: http://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.0020341)
Virulence Factors
Leprosy is a difficult infection to study since it cannot be grown in the laboratory. With recent scientific advancements, more of its infection mechanisms have been brought to light, but many details remain unresolved.
M. leprae begins by infecting Schwann cells found on the exterior of the nerve cells’ axons. The bacterium uses appendages called adhesins to attach itself to the Schwann cells (Figure 2). This results in nerve damage caused by leprosy. In recent years, researchers discovered that M. leprae can accelerate the proliferation of Schwann cell in order to increase the number of cells in which they can infect. Once the host detects a bacterial infection, the immune system initiates the processes that should ultimately lead to the elimination of the invading organism(s). Cells called macrophages are released to phagocytose (or ingest) the bacteria. Normally, the bacteria would be digested within the macrophage, but M. leprae evades cell death using a method that is still not fully understood. M. leprae continues to perpetuate within the macrophage and infects other cells in the host.

Figure 2: The attachment site of M. leprae on Schwann cells (Figure by Emilie Yeh).
Treatment
M. leprae is more difficult to treat than other bacterial infections since it has an abnormally high number of lipid on its cell wall, which acts as a protective barrier against antibiotics from penetrating into the cell. The treatment that is recommended by the World Health Organization are the following multidrug therapies: Dapsone, Rifampin, and Clofazimine. Dapsone inhibits the replication of the cells and all three have bactericidal effects on M. leprae. In the case of tuberculoid leprosy, these antibiotics can reverse heal the patients almost completely; whereas, for lepromatous leprosy, the damage done pre-treatment tend to be irreversible even after the antibiotics have been administered. (Fig. 3) While these drugs have been proven to be extremely effective in curing leprosy, the details on the mechanism of each drug is not yet fully understood.
References
Barker, L. P. (2006). Mycobacterium leprae interactions with the host cell: Recent advances. Indian Journal of Medical Research, 123(6), 748-759.
Hansen’s Disease (Leprosy). Centers for Disease Control and Prevention.
Lastoria J.C., De Abreu M.A.M.M. (2014). Leprosy: Review of the Epidemiological, Clinical, Etiopathogenic Aspects- Part 1. PMC 89(2): 205-218.
Leprosy. World Health Organization. Last updated May 2015.
Leprosy Today. National Institute of Allergies and Infectious Diseases. Last updated February 8, 2011.
Pinheiro R.O., Salles J.D., Samo E.N., Sampaio E.P. (2011). Mycobacterium leprae–host-cell interactions and genetic determinants in leprosy: an overview. NCBI 6(2): 217-230.
Reibel, F., Cambau, E., & Aubry, A. (2015). Update on the epidemiology, diagnosis, and treatment of leprosy. Médecine et Maladies Infectieuses, 45(9), 383-393. DOI: http://dx.doi.org/10.1016/j.medmal.2015.09.002
