Béatrice Bédard-Lepage, Erika Levoy and Rowena Groeneveld
Pseudomonas aeruginosa is a Gram-negative non-fermentative bacterium, meaning it has a thin peptidoglycan layer and is not able to ferment glucose to alcohol. It is an opportunistic pathogen, which means it is usually harmless until the infected person is weakened. P. aeruginosa causes infections difficult to treat in wounds, burns, respiratory tract and bloodstream and was often associated with outbreaks in the past years. In fact, it is responsible for a lot of pneumonia infections acquired in hospitals, because it targets immunosuppressed or artificially breathing patients. Sadly, this pathogen has high rates of morbidity and mortality because it is becoming more and more resistant to multiple antibiotics. For more information on P. aeruginosa check out the article describing this pathogen, (link: https://mechpath.com/2017/12/08/pseudomonas-aeruginosa/).
Usually, we use an antibiotic called aztreonam (Figure 1) to treat P. aeruginosa infection. Aztreonam is a synthetic molecule that contains one 4-membered cyclic amide ring called β-lactam. Unfortunately, many strains of P. aeruginosa are evolving to produce β-lactamase, an enzyme capable of breaking the β-lactam ring in the aztreonam, which cancels the effect of the antibiotic. P. aeruginosa bacteria also have efflux pumps, which are in their membrane and can pump any antibiotics inside the bacteria out before they do any damage. An idea to restore the effect of the antibiotic is to use the aztreonam in combination with another compound, either a β-lactamase inhibitor, or any other molecule that could protect aztreonam or help the antibiotic get inside the bacteria to kill them.
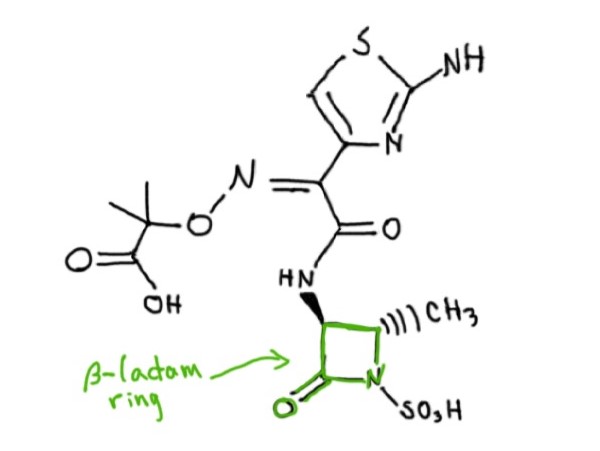
Figure 1: Molecular structure of Aztreonam. Source: Rowena Groeneveld.
The compound that we are interested in today to increase the effects of aztreonam is epigallocatechin gallate, shortened to EGCG (Figure 2). EGCG is a molecule known as a polyphenol and is found in plants that are used to produce tea, such as Camellia sinensis (Figure 3). EGCG is known to be slightly antimicrobial (damaging to bacteria) by itself, but in a recent study, Betts et al., carried out an experiment to determine if EGCG could restore the activity of aztreonam against the resistant strains of P. aeruginosa. They came out with interesting findings in their paper Restoring the activity of the antibiotic aztreonam using the polyphenol epigallocatechin gallate (EGCG) against multidrug-resistant clinical isolates of Pseudomonas aeruginosa and the relevant points of their experiments are discussed below.

Figure 2: molecular structure of EGCG. Source: Rowena Groeneveld.

Figure 3: illustration of C. sinensis. Source: Rowena Groeneveld.
Findings
The first step the scientists took was to look at how much of the antibiotic aztreonam was needed to stop the various strains of P. aeruginosa from growing. Then, they did the same thing with EGCG, and finally they combined the two. They found that with both EGCG and aztreonam combined they needed a lot less of each to stop the bacteria from growing than they would if they used them alone. This means that the two together had a much larger effect than either on its own.
After that, they used the two most antibiotic resistant strains of P. aeruginosa and looked at whether EGCG and the antibiotic could kill the bacteria by seeing if the number of bacteria decreased over 24 hours. Alone, neither was successful, but together they managed to reduce the number of bacteria by a factor of around ten million by the end of the 24 hours.
One thing that was not very clear, was how the EGCG was improving the effect of the antibiotic. The next step the scientists performed was to look at whether EGCG was causing the bacteria to take up, or consume, more of the antibiotic. This would mean more aztreonam can enter the bacterial cell and do more damage before the bacteria can destroy it or get rid of it. They used a fluorescent compound called ethidium bromide and measured how much the pathogen was taking up by itself, and how much it took up when there was EGCG in the mixture. EGCG not only increased the amount of ethidium bromide that was taken up, but also the speed at which it happened.
Now that they could see that EGCG helped the antibiotic destroy the pathogen, they wanted to make sure that it would not damage the host (us). To do this, they grew human skin cells in the presence of EGCG and looked at how many of the cells died. They tested concentrations of EGCG needed to inhibit the growth of 90% of the P. aeruginosa strains and found that after 4 hours at the highest concentration, less than 10% of the human skin cells died.
Next, they did something similar in a species of moth known as Galleria mellonella. However, they also looked at how the antibiotic aztreonam affected the insects, as well as whether the combined effect of EGCG and aztreonam harmed the moths. They found that none of the moths died, even when they used nearly one-hundred times the normal concentration of EGCG. They also checked the moths for melanization. Melanization is an increase of melanine (the skin pigment that allows us to tan to protect ourselves from the UV rays) caused by the immune response of insects. The presence of melanine can be used as an indicator of minor toxicity, and they found no significant increase in melanin for any of the compounds.
After this, they did almost the exact same experiment, but first they infected the moths with P. aeruginosa. They found that when they combined aztreonam with EGCG, a largest amount of moths survived than if they only treated them with aztreonam. This means that the EGCG restores the ability of the antibiotic to kill off the bacteria in the animal model (the moths), but further tests would need to be done to determine if this will work in humans.
Importance of the findings
In a world facing increasing antibiotic resistance, the fact that EGCG can significantly boost the effectiveness of an antibiotic that was once ineffective on its own, comes as a large step in the right direction. What may be even more important, is that although the effectiveness of the antibiotic against P. aeruginosa is increased, the combination with EGCG does not increase the overall toxicity to the human body. This means that this method may be used in the future as a successful form of treatment against P. aeruginosa.
However, P. aeruginosa is not the only bacteria that resists antibiotics nowadays. For example, penicillin resistant strains of Streptococcus pneumonia have appeared, a well-known bacterial pathogen that causes pneumonia and other infectious diseases. The continued threat of antimicrobial resistance will require new ideas, such as the one suggested in this paper, or else humans will once again be threatened by basic bacterial infections.
By continuing to administer unspecific antibiotics, humans may once again live in the age where simple infections like pneumonia could be life threatening.
Despite this, stopping the use of antibiotics is simply not an option in today’s society. Humans have become too dependent on this simple and quick fix to a wide array of infections. Stopping the use of prescribed antibiotics would only slow the progression of microbe’s resistance to antibiotics. It would not provide a solution for the microbes which have already developed resistance. This article proposes a promising solution, wherein antibiotics are still being used, but the period of time in which they are useful is lengthened by combining them with other compounds.
It is novel techniques such as this combination that may allow humanity to stay one step ahead of the ever evolving microbes. Further studies are still needed to discover other combinations of compounds and antibiotics, but if one compound was found in something as common as green tea, it is likely that others are just waiting to be found.
References
Adedeji WA. The treasure called antibiotics. Annals of Ibadan Postgraduate Medicine. 2016;14(2):56–57. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5354621/
Betts JW, Hornsey M, Higgins PG, Lucassen K, Wille J, Salguero FJ, Seifert H, La Ragione RM. 2019. Restoring the activity of the antibiotic aztreonam using the polyphenol epigallocatechin gallate (egcg) against multidrug-resistant clinical isolates of pseudomonas aeruginosa. Journal of Medical Microbiology. 68(10):1552-1559. https://doi-org.proxy3.library.mcgill.ca/10.1099/jmm.0.001060
Buehrle DJ, Shields RK, Clarke LG, Potoski BA, Clancy CJ, Nguyen MH. 2016. Carbapenem-Resistant Pseudomonas aeruginosa Bacteremia: Risk Factors for Mortality and Microbiologic Treatment Failure. Antimicrobial Agents and Chemotherapy. [accessed 2019 Nov 11]; 2016;61(1):e01243-16. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5192105/ doi:10.1128/AAC.01243-16
Osterholm MT, MacDonald KL. Antibiotic-resistant bugs: when, where, and why? Infection Control and Hospital Epidemiology. 1995;16(7):382–4.
[WHO] World Health Organisation. 2017. Global priority list of antibiotic-resistant bacteria to guide research discovery, and development of new antibiotics. [accessed 2019 Nov 11]. https://www.who.int/medicines/publications/WHO-PPL-Short_Summary_25Feb-ET_NM_WHO.pdf?ua=1
Wise R, Hart T, Cars O, Streulens M, Helmuth R, Huovinen P, Sprenger M. 1998. Antimicrobial resistance. BMJ. 317(7159):609. doi: https://doi.org/10.1136/bmj.317.7159.609
