by Wing Chi Cheng and Ramy Elmasry
Introduction
Pertussis, also known as whooping cough (uncontrollable violent coughing), is an infection of the respiratory system originating from the bacterium Bordetella pertussis. It was first isolated and grown in 1906 by Bordet and Gengou. Bordet was later awarded the Nobel prize in Physiology or Medicine in 1920, in part, for his substantial research of B. pertussis as the causative agent of whooping cough. This bacterial pathogen colonizes and multiplies in the upper and lower respiratory tracts. It is highly contagious as it is transferred by coughing or sneezing through airborne droplets.
Disease
Bordetella pertussis primarily targets the respiratory tract as the main site of infection. The infection can be transmitted through coughing or sneezing aerosols. Upon inhalation of contaminated aerosols, the bacteria adhere to the epithelium, which is the protective tissue that surrounds the respiratory airways such as the nasopharynx and trachea. After the attachment process, B. pertussis will start to multiply and produce virulence factors that help the microorganism thrive and cause disease. The collective effect of these factors allows the bacteria to cause damage to the host and elude its immune system.
For instance, they are the main players responsible for the breakdown of cilia found on the respiratory tract (Figure 1). Cilia are membrane protrusions on the surface of respiratory epithelial cells that play a key role in the first line of defense. They mechanically sweep and mediate the clearance of mucus which trap foreign microorganisms and dust particles to be excreted out of the body. Thus, it is the buildup of mucus caused by the deterioration of the cilia that results in the violent coughing. This paroxysmal cough is often followed by a strong gasp for air, which is characterized by the classic “whooping” sound due to narrowed and mucus-filled respiratory airways. Healthier individuals tend to be better at dealing with B. pertussis and display mild symptoms (sneezing, runny nose, low fever) due to their strong immunity.
 Figure 1: Drawing showing the effect of Bordetella pertussis on the ciliated epithelial cells in the respiratory tract. A, normal state of epithelium with regular ciliary function; B, degradation of cilia and breakdown of cells following toxin secretions by B. pertussis. (Source: Wing Chi Cheng)
Figure 1: Drawing showing the effect of Bordetella pertussis on the ciliated epithelial cells in the respiratory tract. A, normal state of epithelium with regular ciliary function; B, degradation of cilia and breakdown of cells following toxin secretions by B. pertussis. (Source: Wing Chi Cheng)
Epidemiology
Over the course of history, Bordetella pertussis has proven to be a very effective pathogen, causing high numbers of morbidity and mortality. Being an endemic disease, pertussis affects regularly developing and developed countries. Sporadic cases also often tend to happen across the globe. This species of Bordetella is human-specific as it is not found in animals or in the environment. Immunocompromised individuals or those with underdeveloped immune systems are more susceptible to having pertussis. This means that newborns and infants under the age of 5 are much more at risk, especially because the last dose of the vaccine against B. pertussis is administered when children are 4-6 years old.
In 2008, there was an outbreak of 16 million cases of whooping cough worldwide, whereby 195,000 were reported dead. Most of the victims were from countries with little access to vaccines. Researches have shown that pertussis differs from other respiratory diseases as it has a higher tendency to cause disease during autumn and winter (seasonally). Even developed countries such as Canada can be affected by this disease. This was the case in 2015 for instance, in Manitoba and New Brunswick where there were 51 and 56 observed cases respectively. These outbreaks are generally due to the presence of unvaccinated people within the community.
Virulence factors
As Bordetella pertussis travels through the respiratory airways, it begins to attach to the protective epithelial cells that line it. It then secretes a series of toxins that damage the host tissues. Toxins are molecules that are synthesized by the bacterium in order to help in its colonization of the host and allow it to evade the immune system.
One of the most important toxins that is specific to this microorganism is the pertussis toxin (PT). Once this toxin is secreted by the bacteria, it binds to the membrane of the host cell and, through special receptors, becomes engulfed by it in a process called endocytosis (Figure 2a). PT then enters the inside of the epithelial cell while enclosed in a bag-like structure called an endosome. A portion of the toxin is then secreted from the endosome and released into the host cell cytosol (Figure 2b) where it begins altering intracellular functions and inflicting damage.
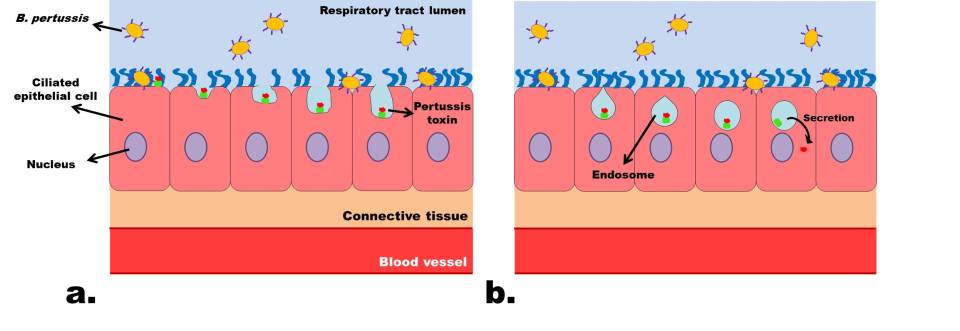
Figure 2: Insertion of the pertussis toxin into host cells. A, attachment to the epithelial cell surface and endocytosis; B, enclosing in endosome and secretion of cytosolic toxin domain. (Source: Ramy Elmasry)
Early in the infection, the pertussis toxin causes a major delay in the recruitment of white blood cells to the site of infection by blocking chemokine production, a signal molecule that is normally sent from the invaded cells to announce danger (Figure 3a). This delay allows the bacteria to multiply and achieve further colonization. It continues by making the host cells hypersensitive to histamine, a compound that promotes leakiness in the vascular membrane (Figure 3b). This leakiness causes the contents of the blood vessels to be absorbed by the tissue below the epithelial cells and causes swelling. This swelling inflicts pressure on the epithelial layer and promotes mucus production, causing the respiratory airways to narrow, making it more difficult to breathe in extreme cases.

Figure 3: Effects of the pertussis toxin on the host system. A, further multiplication and colonization of B. pertussis thanks to a delayed white blood cell response following chemokine inhibition; B, narrowing of respiratory airways caused by mucus buildup and epithelium pressure triggered by the release of blood vessel contents following capillary leakiness after hypersensitivity to histamine. (Source: Ramy Elmasry)
Treatment
B. pertussis is generally susceptible to erythromycin, azithromycin and clarithromycin antibiotics. These help fight against the bacteria by altering or inhibiting protein synthesis, which prevents formation of crucial toxins and virulence factors. These treatments have been proven to be successful in inhibiting B. pertussis and shortening the infection only if administered in the early stages of the disease (first 1-2 weeks).
However, the best way to treat Bordetella pertussis is to prevent it altogether. There are two major types of vaccines that exist that are administered to infants and children at specific ages that have proven to be very successful in prevention: a whole-cell vaccine (DTP) and an acellular vaccine (DTaP). They are named as such because they are prepared as compound vaccines, protecting against Diphtheria (D), Tetanus (T) as well as Pertussis (P) diseases. The whole-cell vaccine, as the name indicates, contains killed whole bacterial cells while the acellular one contains only non-functional virulence factors and inactivated toxins, called toxoids, such as PT. Currently, the favored vaccine that is the most recommended in North America is the acellular format (DTaP) coupled with a booster vaccine (Tdap).
References
Carbonetti NH. 2010. Pertussis toxin and adenylate cyclase toxin: key virulence factors of Bordetella pertussis and cell biology tools. Future microbiology. [accessed 2017 Nov 16]; 5: 455-469. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2851156/. doi:10.2217/fmb.09.133.
1932. The action of histamine on the respiratory tract. The Journal of Physiology. [accessed 2017 Nov 18]; 76. http://onlinelibrary.wiley.com/doi/10.1113/jphysiol.1932.sp002931/abstract. doi: 10.1113/jphysiol.1932.sp002931.
Kilgore, P. E., Salim, A. M., Zervos, M. J., & Schmitt, H. 2016.Pertussis: Microbiology, Disease, Treatment, and Prevention. Clinical Microbiology Reviews. [accessed 2017 November 15]; 29(3): 449–486. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4861987/. doi: 10.1128/CMR.00083-15.
Locht C, Coutte L, Mielcarek N. 2011. The ins and outs of pertussis toxin. FEBS Journal. [accessed 2017 Nov 17]; 278: 4668–4682. http://onlinelibrary.wiley.com/doi/10.1111/j.1742-4658.2011.08237.x/full. doi:10.1111/j.1742-4658.2011.08237.x
Mattoo S, Cherry JD. 2005. Molecular Pathogenesis, Epidemiology, and Clinical Manifestations of Respiratory Infections Due to Bordetella pertussis and Other Bordetella Subspecies. Clinical Microbiology Reviews. [accessed 2017 Nov 15]; 18(2): 326-382. http://cmr.asm.org/content/18/2/326.full?linkType=FULL&resid=18/2/326&journalCode=cmr#sec-20. doi: 10.1128/CMR.18.2.326-382.2005
Melvin JA, Scheller EV, Miller JF, Cotter PA. 2014. Bordetella pertussis pathogenesis: current and future challenges. Nature reviews Microbiology. [accessed 2017 Nov 13]; 12(4): 274-288. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4205565/. doi:10.1038/nrmicro3235.
Zlamy, M. 2016. Rediscovering Pertussis. Frontiers in Pediatrics. [accessed 2017 November 15]; 4:52. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4896922/. doi: 10.3389/fped.2016.00052.
