by Samuelle De Villers-Lacasse and Mélissa Viel
Introduction
Capnocytophaga canimorsus is a bacteria that normally lives in the mouth of dogs and cats without causing symptoms. It retrieves N-acetylglucosamine, a compound it needs for its outer layer, from the components of the saliva of cats and dogs. In humans however, it turns into a pathogen: a microorganism capable of colonizing a host and causing a disease.
Disease
Capnocytophaga canimorsus can be transmitted though bites, licks and being close to animals in general. Symptoms of C. Canimorsus, infections usually appear within 1-8 days after exposure, in most cases around day 2. It feeds on the human host and stays undetected by the immune system. It does not get picked up by the specialized immune cells called macrophages and if by chance it does get picked up, the bacteria can also avoid getting digested by the macrophage. The symptoms after infection may resemble those of the flu but can be as bad as septicemia (blood poisoning). The most common symptoms are fever, vomiting, diarrhea, abdominal pain, confusion, headaches and muscle pain. In more severe cases it can lead to septic shocks, meningitis and peripheral gangrene.
Epidemiology
Cases of C. canimorsus infection have been observed worldwide. In 2005, the Canada Safety Council estimated that dogs bite 460,000 Canadians annually. Between 26-74% of dogs and 18-57% of cats carry C. canimorsus in their mouths. Usually an infection does not happen in healthy individuals but rather high risk ones. These are people with pre existing health conditions, namely alcoholics, people missing their spleen and individuals using steroids . Middle aged and elderly are also more vulnerable to infection. Handling dogs and cats on a regular basis is another risk factor.

Figure 1 Mode of transmission: the presence of dogs or cats with C. canimorsus living in their mouth would bite or lick someone. This would allow the bacteria to make its way into the human causing the symptoms mentioned previously.
Virulence
All human cells have a molecule called sialic acid on their surface to indicate they are from the human host and not from another organism. The sialic acid is on top of a structure of glycan chains (carbohydrates). C. canimorsus secretes an enzyme that cleaves sialic acid on the top and expose the glycan chains that it uses as food ;it can therefore feed on human cells.
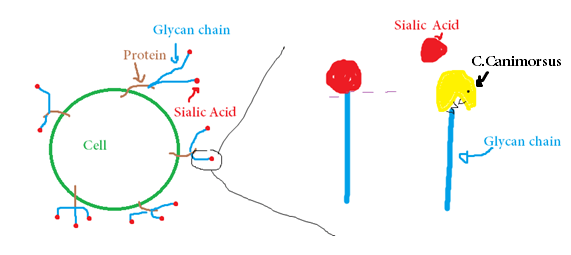
Figure 2: C.canimorsus cleaves the sialic acid on the glycan chains to feed on them.
Normally, macrophages, which are cells of the immune system whose role is to take in and digest unwanted material such as bacteria or cellular debris, would sense C. canimorsus and take it in and kill it by digesting it. .
The problem is, pathogenic strains of C. canimorsus evades the receptor on immune cells that should recognize it :TLR4. TLR4 normally binds to a lipid called lipid A that is a constituent of the outer layer of a group of bacteria called gram negative, because of how they stain under the microscope. C. canimorsus lipid A is different from other gram negative lipid A so won’t interact with TLR4. Non interaction with TLR4 therefore prevents the immune system to recognize that there is a pathogen. Not feeling the threat, TLR4 do not activate an inflammation/recruit immune cells to fight the pathogen, C. canimorsus therefore stays undisturbed.Because it doesn’t bind to TLR4, the microorganism also avoid getting sensed and engulfed by macrophages.
On top of that, the bacteria produces a soluble substance* that prevents the macrophages from killing anything it took in, therefore the bacteria stays in a sac inside the macrophage unharmed. Other bacteria can also benefit from C. canimorsus’ anti-killing protection even if they have been engulfed.
Treatment
The infections with this pathogen are rare but have a high mortality rate. Thankfully there is treatment available when caught early. Usually, after being bitten, the best course of action is to clean the wound. Antibiotics might need to be given if the wound is too deep or if there is a long delay before seeing a medical professional. The antibiotics given, such as penicillin or 3rd generation cephalosporins, usually contain a beta-lactamase inhibitor. Beta- lactamase is an enzyme that the bacteria use to defeat antibiotics. By administering an inhibitor, a molecule that stops the action of beta-lactamase, the bacteria therefore has no defense against those antibiotics.

Figure 3. Beta lactamase prevents antibiotics from working. Beta lactamase inhibitors allows antibiotics to work.
* The specific molecule is still unknown.
References:
1. Meyer, S., Shin, H., & Cornelis, G. (2008). Capnocytophaga canimorsus resists phagocytosis by macrophages and blocks the ability of macrophages to kill other bacteria. Immunobiology, 213(9-10), 805-814. doi:10.1016/j.imbio.2008.07.019
2. Renzi, F., Manfredi, P., Mally, M., Moes, S., Jenö, P., & Cornelis, G. (2011). The N-glycan Glycoprotein Deglycosylation Complex (Gpd) from Capnocytophaga canimorsus Deglycosylates Human IgG. PLoS Pathog PLoS Pathogens, 7(6). doi:10.1371/journal.ppat.1002118
3. Fischer LJ, Weyant RS, White EH and Quinn FD .(1995). Intracellular Multiplication and Toxic Destruction of Cultured Macrophages by Capnocytophaga canimorsus. Infection and Immunity 63 (9): 3484-3490.
4. Pers C, Gahrn-Hansen B, and Frederiksen W. (1996). Capnocytophaga canimorsus Septicemia in Denmark, 1982-1995: Review of 39 Cases. Clinical Infectious Diseases 23: 71-75.
5. Lion C, Escande F and Burdin JC. (1996). Capnocytophaga canimorsus Infections in Human: Review of the Literature and Cases Report. European Journal of Epidemiology 12 (5): 521-533.
6. Shin H, Mally M, Kuhn M, Paroz C and Cornelis GR. (2007). Escape from Immune Surveillance by Capnocytophaga canimorsus. The Journal of Infectious Diseases 195: 375-386.
7. Aggressive Dogs Threaten Public Safety. (2005). Retrieved November 22, 2015, from https://canadasafetycouncil.org/child-safety/aggressive-dogs-threaten-public-safety
8. Gaastra, W., & Lipman, L. (2010). Capnocytophaga canimorsus. Veterinary Microbiology, 140(3-4), 339-346. doi:10.1016/j.vetmic.2009.01.040
9. Mally, M., Shin, H., Paroz, C., Landmann, R., & Cornelis, G. (2008). Capnocytophaga canimorsus: A Human Pathogen Feeding at the Surface of Epithelial Cells and Phagocytes. PLoS Pathog PLoS Pathogens, 4(9). doi:10.1371/journal.ppat.1000164
10. Jolivet-Gougeon A, Sixou JL, Tamanai-Shacoori Z, Bonnaure-Mallet M. (2007). Antimicrobial treatment of Capnocytophaga infections. Int J Antimicrob Agents 2007;29:367–373.
11. Watson ID, Stewart MJ, Platt DJ (1988). “Clinical pharmacokinetics of enzyme inhibitors in antimicrobial chemotherapy”. Clin Pharmacokinet 15 (3): 133–64. doi:10.2165/00003088-198815030-00001. PMID 3052984.
12.Weese, S., & Anderson, M. (2013, August 1). Capnocytophage for pet owners. Retrieved November 22, 2015, from http://www.wormsandgermsblog.com/files/2008/04/M1-Capnocytophaga.pdf
