by Leah Assouline and Jessica Breitstein
Introduction
Helicobacter pylori, has infected humans for more than 58000 years but was only discovered and isolated by two australians, Barry J. Marshall and Robin Warren in 1982. This pathogen was proven to cause ulcers, which was previously linked to stress and eating certain food. Interestingly, Barry J. Marshall, desperate to prove the connection between H. pylori and ulcers, ingested H. pylori taken from the gut of a sick patient and, indeed, developed ulcers.
Disease
The method of transmission of Helicobacter pylori is still unknown. It is believed that it transmitted from human to human through oral or fecal matter. In the stomach, H. pylori will infect gastric epithelial cells after passing through the mucosal layer. This species of bacteria has adapted to survive in the acidic environment of the gut (Figure 1). It is able to modify the host cell’s physiology and undermine the immune system to endure inside the host through its lifetime. Without any intervention, the diseases that are caused by this pathogen range from chronic gastritis with no symptoms, to peptic ulcers, to severe gastric cancer. Furthermore, Helicobacter pylori is the first bacteria to be associated with cancer.
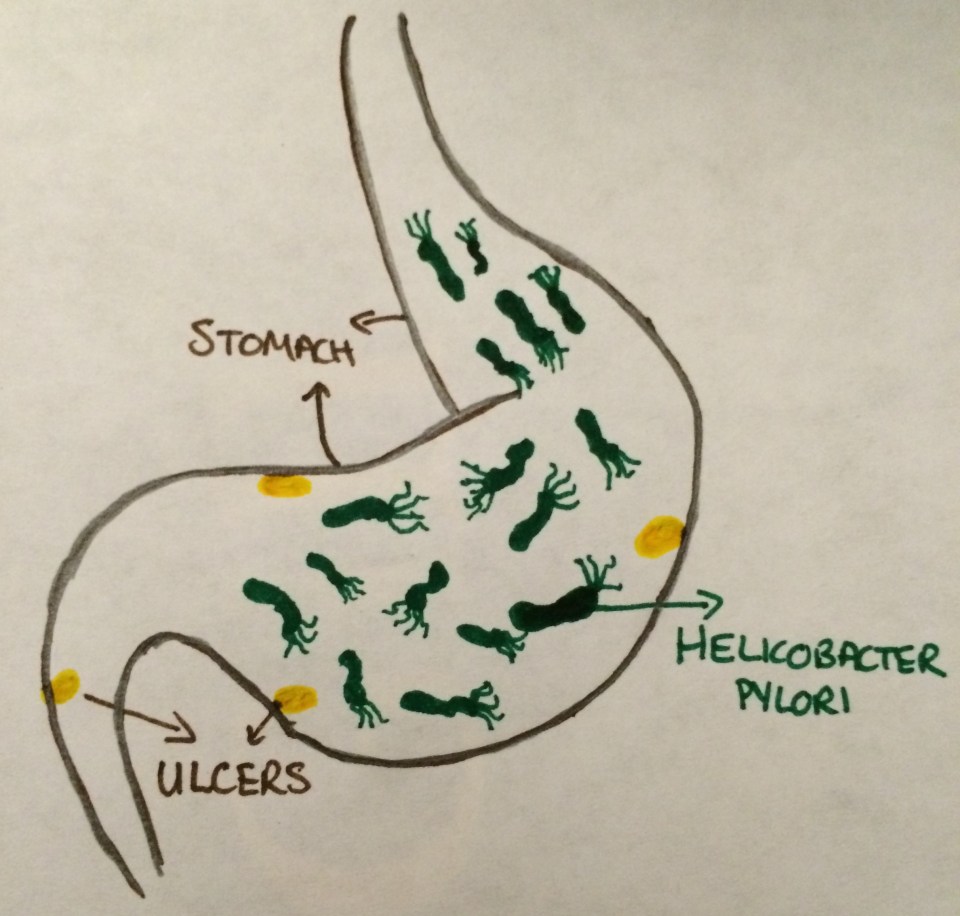
Figure 1: Ulcer formation caused by Helicobacter pylori infection in human stomach.
Epidemiology
It is shown in multiple studies that over half of the world’s population is infected with H. pylori. In developing countries, the prevalence of H. pylori infection is higher due to the lower sanitation standards, and due to contaminated drinking water. Studies show that H. pylori infection causes 90% of all duodenal ulcers and 80% of all stomach ulcers. Indeed, it is seen that each year there are over 800,000 new cases of peptic ulcers. Furthermore, long term infection of H. pylori is known to cause gastric cancer, which is the second most common cancer worldwide.
Virulence factors
Since H. pylori thrives in the host stomach, a highly acidic environment, it needs to protect itself from the acidity. All H. pylori strains do this by expressing a urease enzyme (Figure 2). A urease will break down the urea in the stomach, a byproduct produced in the liver from breakdown of amino acids, into ammonia and carbon dioxide. The ammonia will neutralize the acidity surrounding the bacteria, allowing it to migrate to the mucus layer bordering the gastric epithelial cells. The mucus will usually protect epithelial cells from bacteria but the ammonia will solubilize the mucus, allowing the H. pylori to cross to the epithelial cells.
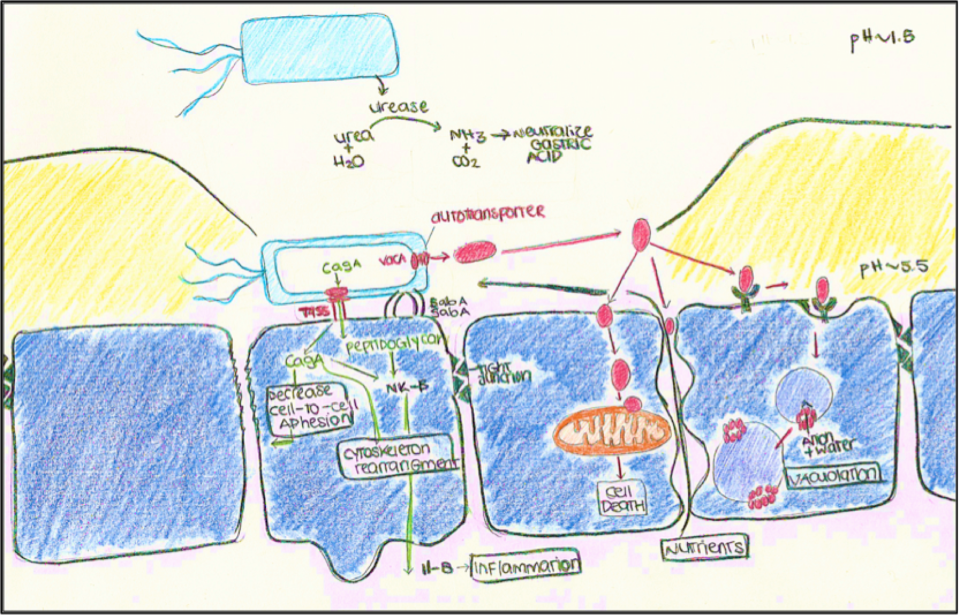
Figure 2: Function of Urease, adhesion of H. Pylori by BabA/SabA, T4SS secretion of cagA and its function and the function of the autotransporter VacA.
Once H. pylori are through the mucus layer, it will adhere to the host’s epithelial cell membrane through adhesins, SabA and BabA, outer membrane proteins, allowing a delivery system for virulence factors to bind to the host cell (Figure 2). The Type 4 secretory system (T4SS) in Helicobacter pylori, has an inner and outer channel forming needle-like projection, to facilitate contact between bacteria and host. After contact with host cell, H. pylori will use many different virulence factors with different functions, allowing it to survive and colonize the host cells (Figure 2).
One of the virulence factors is the CagA protein injected by the T4SS. Its function is to decrease cell-to-cell adhesion and rearrange the cells cytoskeleton. The T4SS will also inject peptidoglycan into cell, which will, with CagA trigger the release of a chemokine, il-8, a signalling protein that triggers persistent inflammation as the immune response and potentially causes the ulcers. Furthermore, CagA will affect cell survival after infection, which can lead to the development of gastric cancer.
Another important virulence factor is the VacA protein. It will, unlike the CagA, form an autotransporter in H. pylori membrane to secrete itself. VacA binds to receptor on host cells, triggering the cell to engulf it into vacuoles. The vacuole, or storage bubbles, forms channels in its membrane, allowing protons to flow inside, as well as water, leading to increased acidity and swelling. VacA will also insert inside the mitochondria, leading to the cell death. Finally, as seen in Figure 2, VacA will disrupt the barrier of the cells, allowing the escape of nutrients to the H. pylori and improving its survival in the stomach.
Treatment
Multiple antibiotics can be used for the treatment of H. pylori infection. Common antibiotics that are used include clarithromycin, amoxicillin, metronidazole, tetracycline, and fluoroquinolones. Proton pump inhibitors, which lower acidity in the stomach, and bismuth products are also frequently added to the treatment therapy. For effective treatment, infected individuals will take a combination of two or more antibiotics for a two week period. These antibiotics can each clear the infection of H. pylori in a different way. For example, clarithromycin blocks the bacterial protein synthesis and amoxicillin interacts with the bacterial cell wall to weaken it and cause bacterial cell death.
References
Brown, L. M. (2000). Helicobacter Pylori: Epidemiology and Routes of Transmission. Epidemiologic Reviews. 22.2, 283-97.
Wandler, A., et al. (2010). A Greasy Foothold for Helicobacter pylori. Cell Host & Microbe 7.5, 338-339.
Sutton, P., et al. (2010). Helicobacter Pylori in the 21st Century. Wallingford, Oxfordshire.
Veiga, N., et al. (2015). Oral and Gastric Helicobacter Pylori: Effects and Associations. PLOS ONE, 10.5.
Mégraud, F. (2005). A Humble Bacterium Sweeps This Year’s Nobel Prize. Cell 123.6, 975-76.
Testerman, T.L. (2014). Beyond the Stomach: An Updated View of Helicobacter Pylori Pathogenesis, Diagnosis, and Treatment. World Journal of Gastroenterology 20.36, 12781.
Posselt, G., et al. (2013). The Functional Interplay of Helicobacter Pylori Factors with Gastric Epithelial Cells Induces a Multi-step Process in Pathogenesis. Cell Communication and Signaling Cell Commun Signal 11.1, 77.
Cellini, L., et al. (2000). Virulence Factors of Helicobacter Pylori. Microbial Ecology in Health & Disease 12.2, 259-62.
Adler, I., et al. (2014). Helicobacter Pylori and oral pathology: Relationship with the gastric infection. World Journal of Gastroenterology. 20.29, 9922–9935.
