By Elena Lonina, Xinlan Yang and Yueran Zhao,
Introduction
Chlamydia pneumoniae, also known as Chlamydophila pneumoniae, is a human pathogen that causes acute respiratory diseases such as pneumonia and bronchitis. It is an obligate intracellular parasite, which implies that it needs to reside inside a host cell to grow. C. pneumoniae was first isolated in 1965 but was not proved to be a pathogen until 1983. School-age children are the most susceptible target and carrier of the bacteria, with mild or even no symptoms following the infection. Chlamydia infections are known to be community-acquired, which means that the disease can spread in a community if one individual is sick. Therefore, re-infections are common in the older populations given that they can be easily exposed to the pathogen in their daily lives and their symptoms may be more severe due to the existence of co-infecting bacteria or underlying diseases.
Disease
C. pneumoniae infections are associated with severe systemic infections and coronary artery and acute respiratory diseases. It is transmitted between human carriers with or without symptoms through secreted droplets from the respiratory system, such as by sneezing or coughing. C. pneumoniaemay incubate inside the human body for several weeks since its entry to the onset of symptoms. C. pneumoniae is biphasic, which means it has two forms during its life cycle: the elementary body (EB), which has decreased intracellular activity to minimize energy consumption, and the reticulate body (RB) which is metabolically active and engages in replication. Further, the bacteria are infectious as EB but are non-infectious as RB. Although the bacteria require obligate intracellular growth, the EB form can survive extracellularly while waiting to reach a new host. These EB are picked up by immune cells called macrophages (Figure 1), which are recruited when the host cells detect a bacterial invasion, and start working to take up the intruder within little sacs called vesicles. Inside of the vesicles, C. pneumoniae blocks the fusion of the vesicle they reside in to lysosomes, which carries deadly enzymes to the bacterium, to prevent being killed. Then, the EB matures to an RB inside the hijacked vesicle and starts its replication process. This bacterial infection cannot be characterized by a specific set of syndromes, but common symptoms include sore throat and loss of voice before it develops into pneumonia or bronchitis. Even when the infection appears to be mild, the clearing of the bacteria from the patients’ bodies appears to be slow.

Figure 1. Transmission electron micrograph showing Chlamydia pneumoniae’s Elementary Bodies (arrows) attaching to the surface of the macrophage (A) and, once inside the macrophage, they attach to the surface of the vesicles before differentiation into reticular bodies and replication (B). Source: Hahn et al., 2002.
Epidemiology
C. pneumoniae is one of the most common pathogens that cause upper and lower respiratory tract infections. As mentioned before, this bacterium can be easily transmitted from human to human via tiny droplets spread over in the air. When someone has a C. pneumoniae infection within a community with high population density, the risk of infection becomes higher because there would be a higher number of potential recipients that could catch the pathogen, and thus the probability of a C. pneumoniae outbreak would be increased.
C. pneumoniae causes infections in people of all ages globally. Noticeably, there is a distinctive characteristic of this infectious disease, which is that patients who have been infected by C. pneumoniae recently are more susceptible to a reinfection. Moreover, the susceptibility of an individual is greatly impacted by a series of factors. To illustrate, research has shown the most frequent ages that people get the very first C. pneumoniae infection is within a range of 5-15. This is most likely due to the underdeveloped immune systems of children and teenagers. Other conditions such as aging, obesity, immunocompromising conditions and underlying diseases (such as AIDS and chronic pulmonary diseases) also increase susceptibility, especially in adults. These underlying conditions usually make reinfections more dangerous for adults.
Virulence factors
Given that C. pneumoniae is an obligate intracellular pathogen, its internalization into host cells is crucial for its maturation, replication, and dissemination – events that will generate host damage and define the disease. Nevertheless, any incoming pathogen will need to face the first mechanical barrier that will limit its translocation and invasion: the plasma membrane. Thus, it is not surprising that C. pneumoniaehas evolved different methods to invade its host and ensure its survival.
One example is OmcB, an adhesin (a molecule that mediates adhesion) that has shown to attach to specific molecules present only in host cell membranes. What is surprising, however, is that OmcB can have little differences in its structures and that these will allow for C. pneumoniaeto bind to different variants of these host molecules that are spread throughout different types of cells, conferring specificity to its attachment.
C. pneumoniae is even further able to induce more complex changes that will enhance its internalization. To illustrate, Pmp21 is also an adhesin, but this one binds to a receptor present in the host cell membrane. When the adhesin binds, it activates the receptor and signals the cell to change its structure and to extend arms around the pathogen binding to the receptor (Figure 2). This way, C. pneumoniae actively stimulates the cell to take it in.
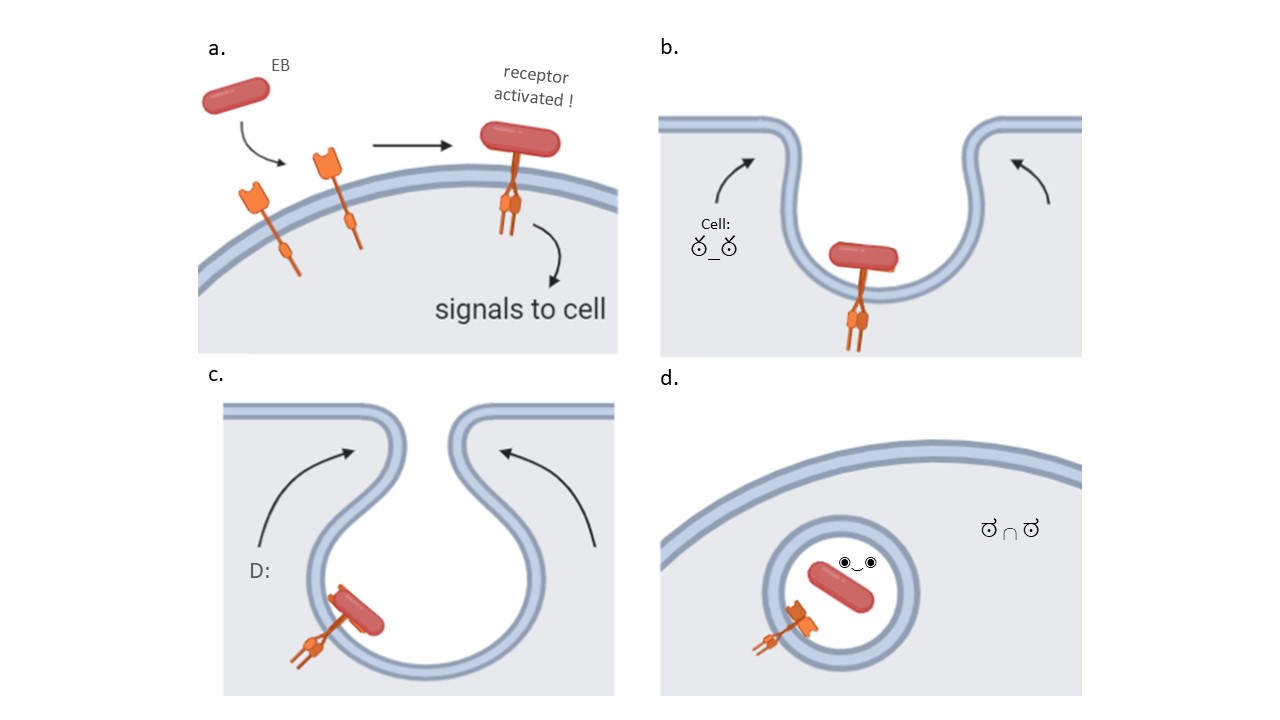
Figure 2. Chlamydia pneumoniae binds to specific host membrane structures to begin the invasion. For example, some adhesins in the Elementary Body (EB) can bind to host receptors located in its plasma membrane and activate it (a). Receptor activation signals the cell to start extending arms around the receptor and bringing it inwards (b and c). Ultimately, a vesicle forms with the bacterium inside of it, where it can mature and start replicating (d). Source: Elena Lonina.
Another example of active stimulation is Cpn0473, which is an adhesin expressed in adhering EB and that binds preferably to cholesterol-rich regions in the host’s cell plasma membrane. What is interesting, however, is that the insertion of Cpn0473 in the plasma membrane will change the molecular composition of the plasma membrane. This change is usually associated with apoptosis of the host cell. However, the change initiated by Cpn0473 does not induce cell death, but instead induces endocytosis of the bacterium, which results in the internalisation of the bacterium similarly to what is observed in Figure 2.
Treatments
Antibiotics are the optimal treatments for bacterial infections. Clinically, erythromycin and doxycycline are established treatments that can effectively clear the infection by inhibiting bacterial protein synthesis, which means that it will not be able to create the molecules that will allow it to attach to the host, invade it, and mature. However, there are several side effects associated with these antibiotics, such as diarrhea, vomiting and headache. Other antibiotics such as tetracyclines and ofloxacin are applicable to adults, but they are restricted to children because of their potential to cause damage to their growing muscles, tendons, and skeletons. Usually, due to its high reinfection rick as mentioned above, a relatively large dose and long course of treatment is required to ensure the complete clearance of the pathogens.
References
Blasi, F., Arosio, C., and Cosentini, R. 1999. Chlamydia pneumoniae: Epidemiology. In Chlamydia pneumoniae: The Lung and the Heart. Edited by L. Allegra and F. Blasi. Springer Milan, Milano. pp. 52-61.
Fechtner, T., Galle, J.N., and Hegemann, J.H. 2016. The novel chlamydial adhesin CPn0473 mediates the lipid raft-dependent uptake of Chlamydia pneumoniae. Cell Microbiol 18(8): 1094-1105. doi:10.1111/cmi.12569.
Galle, J.N., Fechtner, T., Eierhoff, T., Römer, W., and Hegemann, J.H. 2019. A Chlamydia pneumoniae adhesin induces phosphatidylserine exposure on host cells. Nature Communications 10(1): 4644. doi:10.1038/s41467-019-12419-8.
Hahn, D.L. 2002. Chlamydia pneumoniae as a respiratory pathogen. Frontiers in Bioscience 7(1-3): e66. doi:10.2741/hahn.
Kuo, C.C., Jackson, L.A., Campbell, L.A., and Grayston, J.T. 1995. Chlamydia pneumoniae (TWAR). Clinical Microbiology Reviews 8(4): 451. doi:10.1128/CMR.8.4.451.
Moelleken, K., and Hegemann, J.H. 2008. The Chlamydia outer membrane protein OmcB is required for adhesion and exhibits biovar-specific differences in glycosaminoglycan binding. Molecular Microbiology 67(2): 403-419. doi:10.1111/j.1365-2958.2007.06050.x.
Mölleken, K., Becker, E., and Hegemann, J.H. 2013. The Chlamydia pneumoniae Invasin Protein Pmp21 Recruits the EGF Receptor for Host Cell Entry. PLOS Pathogens 9(4): e1003325. doi:10.1371/journal.ppat.1003325.
Paldanius, M., Bloigu, A., Alho, M., Leinonen, M., and Saikku, P. 2005. Prevalence and Persistence of <em>Chlamydia pneumoniae</em> Antibodies in Healthy Laboratory Personnel in Finland. Clinical and Diagnostic Laboratory Immunology 12(5): 654. doi:10.1128/CDLI.12.5.654-659.2005.
