By: Bérénice Saget, Jess and Emilia
This article is to go further after the bacterial pathogen Borrelia burgdorferi post, go check it out!
Climate change is a recurring theme in today’s media. We hear and read about how our planet is warming, how the climate is becoming more unpredictable, and how this is negatively impacting our lives. Amidst all of this, there is a tiny organism that is greatly benefiting. Meet Ixodes scapularis, better known as the black-legged tick, and its friend, Borrelia burgdorferi, a pathogenic bacterium it can carry. These names might be familiar to you in the context of Lyme disease, which has been on the rise in the Northern hemisphere in recent years.
Lyme Disease
Lyme disease is caused by an infection with the pathogenic bacterium, B. burgdorferi. When a tick attaches and bites through an individual’s skin, the pathogen can be transmitted with the tick’s saliva.
Signs & Symptoms
The first characteristic sign is the “bull’s eye”, also known as erythema migrans rash. This will appear at the site of the tick bite. In the early stages of the disease, a wide variety of symptoms may manifest including but not limited to:
- severe headaches
- facial paralysis
- muscle & bone aches
- heart disorders
- memory loss
- nerve pain
- arthritis
The disease is said to be in the early localized stages in the first month post-exposure as the infection stays confined in the skin at the site of the bite. In the early disseminated stage, the bacterium has migrated to the bloodstream and colonized other sites of the body. The disease is classified as at a later stage when the symptoms have persisted for several months to years. It is unclear why symptoms persist after the infection has been cleared. However, it has been proposed that lasting symptoms are caused by the antibodies created during infection that attack the body’s cells that resemble the bacterium’s antigens, leading to an autoimmune disease. Symptoms can become more severe if the infection is left untreated. In rare cases, Lyme disease can lead to death from complications if the heart becomes infected.
Interaction between B. burgdorferi and its vector
Borrelia burgdorferi is a Gram-negative bacterium that grows very slowly. Many different B. burgdorferi strains are associated with Lyme disease and their vector.
The Borrelia genus is very vast and the different species are hard to differentiate without sequencing the genome. The bacteria within a species also have high variability meaning many different B. burgdorferi strains are associated with Lyme disease.
This genus is quite unique in the way its metabolism functions. It has evolved to use manganese instead of iron in all of its metabolic pathways. This allows the bacteria to grow in our blood, where the iron is bound to heme and other proteins, without having to synthesize iron-acquiring complexes. The bacteria waste less energy and can focus on escaping the immune system.
Borrelia spp. uses different techniques to hide in its host and evade the immune system. This allows it to survive longer and gives it a better chance to spread by being transported to another host by a tick.
The environmental biology of this pathogen is complex since it has a broad spectrum of mammals, birds, and reptiles it can infect. It is vehiculated between hosts by various tick vectors. Many ticks can carry Borrelia spp., and are perfect vectors due to the long-lasting blood meals their bodies can sustain and the enzymes of their saliva preventing inflammation and blood clotting at the infection site. To evade our immune system, the bacteria will increase production of those salivary proteins in infected ticks and bind them.
Coevolution of the ticks and their bacterial guests made them tolerant to one another. The immune system of the tick does not kill the bacteria entirely, it only keeps it in check. Once inside a tick, Borrelia increases the amount of its OspA surface proteins to mediate adhesion to the tick’s gut. There, it remains attached and grows using the nutrients from the tick’s blood meal. Once the tick has digested the blood, Borrelia will change OspA for OspC, which permits its migration to the tick’s vascular system. It will then travel to the salivary glands to be secreted into the tick’s new host.
The bacteria has adapted very well to the tick, using its nutrients and body as a means of transport without disturbing it.
The tick’s life cycle being complex, the bacteria need to adapt quickly to survive during molting and the passage from tick to mammal. This change of host is sensed by the temperature increase and causes changes in the gene expression of Borrelia to adapt to the change in environments.
Effect of Climate Change on the Tick Vector
For years, scientists have been warning the public that the Earth is warming and that this could have detrimental environmental and economic effects on life as we know it. Worldwide, there has been an alarming increase in temperature, precipitation patterns, and extreme weather events that are all associated with climate change. The local ecology has not been impervious to these effects either. In Canada alone, there has been documented evidence that increases in temperature have influenced ticks. Hotter temperatures have promoted better conditions for ticks to survive and accelerate their lifecycle, leading to faster propagation.
The Tick’s life cycle
The tick’s life cycle involves 4 stages: eggs, larva, nymph, and adult (Figure 1). Nymphal and adult stages are the infective ones to humans although every stage requires a blood meal from different hosts. Every blood meal gives the pathogen a chance to be transmitted, resulting in spreading of the bacterium as well as the molting or mating of the tick. Humans, however, do not permit the reproduction of the ticks nor B. burgdorferi.
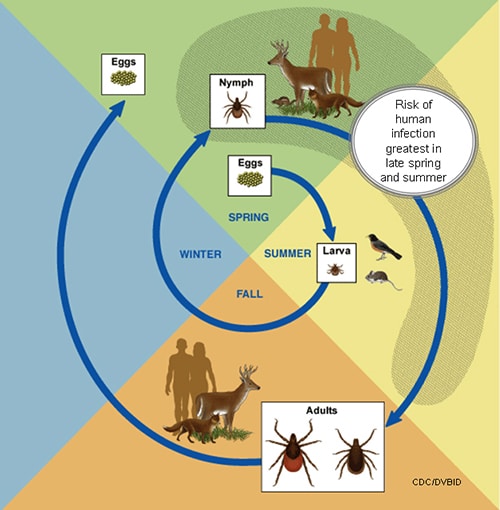
Hotter temperatures result in a faster developmental cycle, increased egg production and thus, an increased population. Once larvae have molted into the nymphal stage, the cold winter forces them to remain dormant until spring. However, with warmer winters, these ticks no longer need to hibernate and can become active on warmer days. Warmer winters also affect the tick’s reservoir, especially the white-footed mouse. The mice reproduce in the spring and summer, meaning that shorter winters will also yield higher rates of the tick’s host.
All these changes are permitting the tick population to grow along with its reservoirs and increases their activity even during the winter. Milder temperatures are enabling them to spread to more northern latitudes. Additionally, longer spring and fall seasons have led to humans spending more time outdoors and wearing summer clothing for longer, which also increases the likelihood of tick exposures and therefore, Lyme disease.
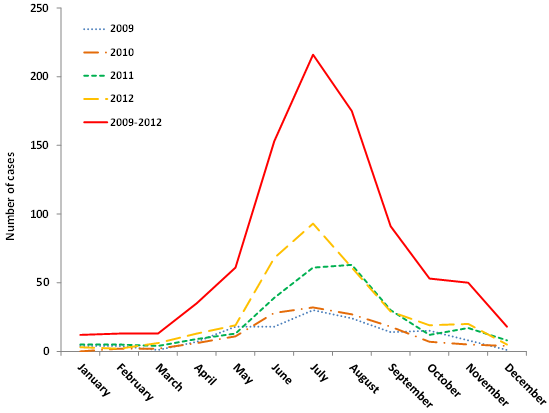
According to the CDC, the number of confirmed cases of Lyme disease has risen from 12,801 cases in 1997 to 29,513 in 2017. A similar trend is observed in Canada (Figure 2). As Lyme disease is becoming more prevalent, it has been receiving increased media coverage and attention from public health agencies. Through education about the disease and its route of transmission, we can promote increased vigilance when outdoors and catch infections early. While climate change threatens to increase the prevalence of the disease, prevention of Lyme disease by education and vaccination will promote better outcomes.
Vaccines & Prevention
Even though the bacteria show signs of resistance to erythromycin, they are still susceptible to most antibiotics. However, this may change in the future. The more the bacteria is exposed to antibiotics and courses of antibiotic treatment are not completed, the higher the chances of developing antimicrobial resistance (AMR).
In an effort to use fewer antibiotics, a vaccine was created in the US to decrease prevalence of Lyme using the surface Osp A protein as a template to create antibodies. While the vaccine proved to be effective, an unfounded uproar from the public caused widespread disapproval of the vaccine, which was consequently discontinued. A new vaccine VLA15 based on six different variations of the same Osp A protein is currently in the second phase of clinical trials and shows promising results according to the FDA.
The current best practices to prevent the spread of Lyme disease is avoidance of tick-infested areas. If these areas must be visited, skin should be covered by long pants and sleeves; wearing light colors is helpful in spotting the insect. Insect repellents containing DEET are also useful.
References
Barbour, A., & Garon, C. (1988). The genes encoding major surface proteins of Borrelia burgdorferi are located on a plasmid. Annals of the New York Academy of Sciences, 539(1 Lyme Disease), 144-153. doi:10.1111/j.1749-6632.1988.tb31847.x
Bednar, T. (2018, October 9). What Will Climate Change Mean for Lyme Disease? Retrieved October 22, 2019, from https://serc.carleton.edu/NAGTWorkshops/health/case_studies/lyme_disease.html.
Bouchard, C., Dibernardo, A., Koffi, J., Wood, H., Leighton, P., & Lindsay, L. (2019). Increased risk of tick-borne diseases with climate and environmental changes. Canada Communicable Disease Report = Releve Des Maladies Transmissibles Au Canada, 45(4), 83-89. doi:10.14745/ccdr.v45i04a02
Bowman, A., & Nuttall, P. (2008). Ticks : Biology, disease and control. Cambridge, UK: Cambridge University Press.
Centers for Disease Control and Prevention. (2018, December 21). Lyme Disease Charts and Figures: Historical Data. Retrieved October 22, 2019, from https://www.cdc.gov/lyme/stats/graphs.html.
Comstedt, P., Schüler, W., Meinke, A., & Lundberg, U. (2017). The novel Lyme borreliosis vaccine vla15 shows broad protection against Borrelia species expressing six different ospA serotypes. Plos One, 12(9), 0184357. doi:10.1371/journal.pone.0184357
De, T., Kreuk, L., Van, D., Hovius, J., & Schuijt, T. (2013). Complement evasion by Borrelia burgdorferi: It takes three to tango. Trends in Parasitology, 29(3), 119-28. doi:10.1016/j.pt.2012.12.001
Fallon, B.A., & Sotsky, J. (2018). Conquering Lyme disease : Science bridges the great divide. New York: Columbia University Press. (2018).
Gray, J. (2002). Lyme borreliosis : Biology, epidemiology, and control. Wallingford, Oxon, UK: CABI Pub.
Hajdusek O., Sima, R., Ayllon N., Jalovecka M., Perner J., De La Fuente J., Kopacek P. (2013). Interaction of the tick immune system with transmitted pathogens. Frontiers in Cellular and Infection Microbiology, 3(26), 2235-2988. Doi: 10.3389/fcimb.2013.00026
Hu, L. (2016). Lyme disease. Annals of Internal Medicine, 164(9), 80. doi:10.7326/AITC201605030
Jackson, C., Boylan, J., Frye, J., & Gherardini, F. (2007). Evidence of a conjugal erythromycin resistance element in the Lyme disease spirochete Borrelia burgdorferi. International Journal of Antimicrobial Agents, 30(6), 496-504. doi:10.1016/j.ijantimicag.2007.07.013
Jewett, M., Lawrence, K., Bestor, A., Tilly, K., Grimm, D., Shaw, P., VanRaden, M., Gherardini, F., Rosa, P. (2007). The critical role of the linear plasmid lp36 in the infectious cycle of Borrelia burgdorferi. Molecular Microbiology, 64(5), 1358-74.
Nigrovic, L.E., Thompson, K.M. (2007). The Lyme vaccine: A cautionary tale. Epidemiology and Infection, 135(1), 1-8.
Patterson, B. (2015, May 4). Global Warming May Spread Lyme Disease. Retrieved November 8, 2019, from https://www.scientificamerican.com/article/global-warming-may-spread-lyme-disease/.
Petnicki-Ocwieja, T., Brissette, C. A. (2015). Lyme disease: recent advances and perspectives. Frontiers in cellular and infection microbiology, 5, 27. doi:10.3389/fcimb.2015.00027
Public Health Agency of Canada. (2017, June 13). Government of Canada. Retrieved from https://www.canada.ca/en/public-health/services/diseases/lyme-disease/symptoms-lyme-disease.html.
Ras, N., Postic, D., Ave, P., Huerre, M., & Baranton, G. (2000). Antigenic variation of Borrelia turicatae vsp surface lipoproteins occurs in vitro and generates novel serotypes. Research in Microbiology, 151(1), 5-12. doi:10.1016/S0923-2508(00)00133-9
