by Jade Lee
Introduction
Burkholderia pseudomallei also known as Pseudomonas pseudomallei is a gram-negative bacterium that causes Melioidosis or Whitemore’s disease. It can grow on a variety of media and forms wrinkled, pinkish colonies (see figure 1). B. pseudomallei exits as a saprophyte in wet soils and rice paddies but is capable of infecting humans and many animal species (including sheep, goats, horses, swine, cats, dogs, cattle). A saprophyte is an organism which gets its energy from dead and decaying organic matter.
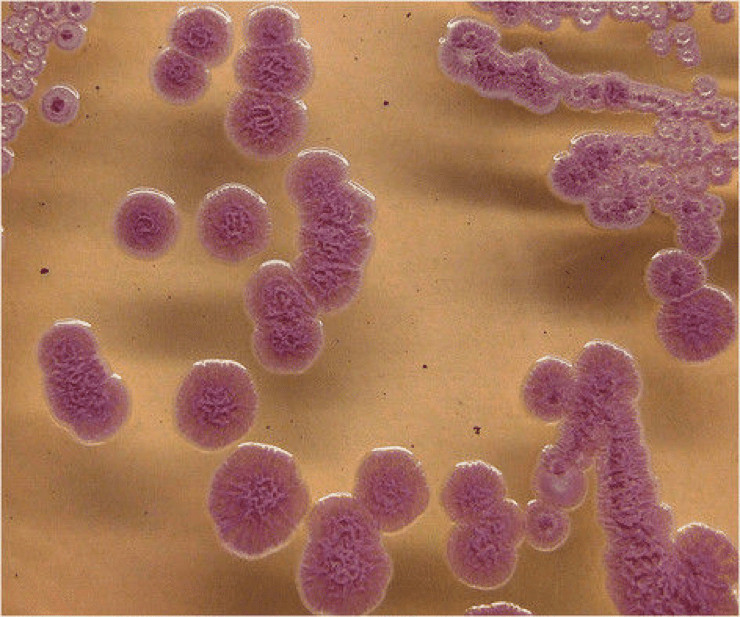
Figure 1: Colonies of B. pseudomallei on Ashdown’s agar showing the typical pink crinkled colonies after 72 hours of incubation at 37°C. Source: BioMed Central Veterinary Research, 2014.
Disease
Disease occurs when the bacteria enters the body via cuts and sores in the skin or via inhalation of dust or droplets and very rarely by ingestion of contaminated water. Direct human-to-human and animal-human transmission is rare but may occur after contact with blood or bodily fluids. Indeed, infected animals and humans can shed B. pseudomallei in urine, feces, wound exudates, nasal secretions and milk, these secretions will depend on the site of infection. B. pseudomallei is capable of remaining latent for up to 29 years (longest documented period of latency) because it is a facultative intracellular pathogen and can escape the host’s immune system by invading and multiplying in phagocytic and nonphagocytic cells. Phagocytic cells (neutrophils, monocytes and macrophages) are cells of the immune systems whose role is to take up and digest invading pathogens. The nonphagocytic cells that the bacteria can invade are epithelial cells; epithelial tissues are thin tissues that cover all the exposed surfaces of the body. The bacterial invasion of these cells can result in their lysis. Melioidosis causes localized disease, such as pneumonia and abscesses. The symptoms are non specific: fever (high grade), headache, vomiting, nausea abdominal pain, cough, and lung abscess. B. pseudomallei can also spread to secondary sites, including organs such as liver, spleen or brain, or to the blood, resulting in septicaemia (blood poisoning). The infection can also be asymptomatic.
Epidemiology
Melioidosis is primarily a disease of tropical climates, specifically found in Southeast Asia and northern Australia. B. pseudomallei was recovered in 20.4 % of soil samples in northeastern Thailand and accounts for 20% of community-acquired (infection contracted outside of a health care setting) blood poisoning or septicemias in this region where 50% of those affected die. It usually acts as an opportunistic pathogen, meaning that it can infect a host when the host’s resistance is compromised. Indeed, people that are usually at risk for Melioidosis are those with conditions such as diabetes, kidney disease, lung disease, cancer, heavy alcohol consumption and those on immunosuppressive therapy including steroids. However, healthy people can acquire the disease if they work in muddy soil without adequate hand and foot protection. Children are usually less likely to get the disease than adults but they can still get it during the wet season, especially those with a weakened immune system.
Virulence systems
Initial infection occurs at the epithelial cell layer of either the wounded skin or the mucosal surface. The attachment to these cells is mediated by a thin polysaccharide layer around the bacteria or capsule, and via a type IV pilus. Type IV pili are strong, flexible filaments that are like grappling hooks that can attach to the surface of other cells, and while they retract, they pull the bacterium towards the point of attachment. The capsule of B. pseudomallei can also help the bacterium evade the host’s immune system by resisting phagocytosis. Once the epithelium cells or phagocytic vacuoles are invaded, B. pseudomallei is able to escape from endocytic vacuoles or phagosomes (membrane-bound cellular organelles that engulfed the bacteria) via the type III secretion called bsa. This is a syringe like molecule that injects toxins through the wall of the membrane of the host cell. These toxins enable the bacteria to escape the phagosome and inhibit autophagy, a process that allows host cells to deal with intracellular infection by packaging, degrading and recycling cellular contents. B. pseudomallei is then able to spread from cell to cell by recruiting to its surface host actin and other proteins present in the host cells, which initiates the assembly of an actin tail. The continous assembly of the actin tail supplies force to propel the organism through the cytoplasm of the infected cell and into neighbouring cells (see figure 2). In phagocytic cells B. pseudomallei is also able to neutralize reactive oxygen species and proteases (enzymes that degrade proteins) that normally destroy the pathogens. Finally, the pathogen can also produce haemolysins (which destroy red blood cells), lipases (enzymes that destroy lipids) and siderophores (complexes that have the ability to sequester iron from host cells). These secreted factors have cytotoxic effects on eukaryotic cells and cause further damage to the host’s tissues.
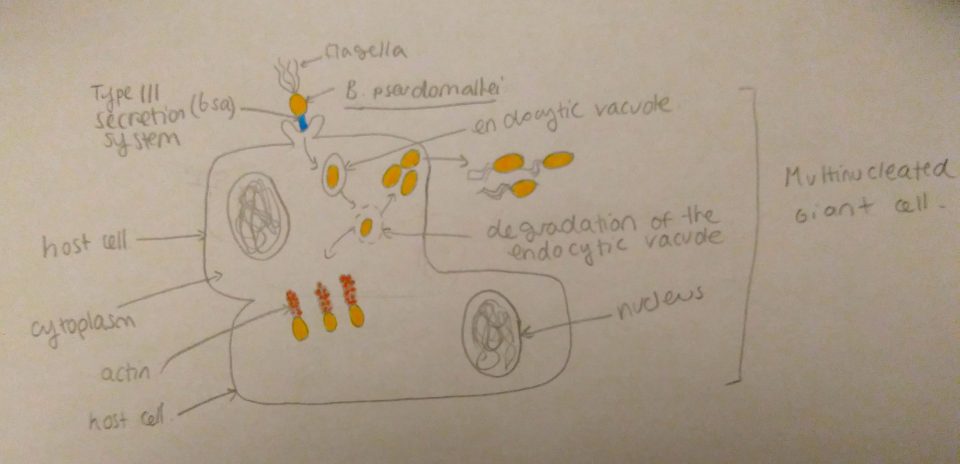
Figure 2: Invasion of host cells by B. pseudomallei: upon entry in the host cell, the bacteria lyses the phagocytic vacuole with its Type III secretion system, once in the cytoplasm the bacteria assembles an actin tail at one of its poles, this assembly propels the bacteria to other adjacent cells. Source: Jade Lee (2017).
Treatment
Antibiotics are less effective against B. pseudomallei because of its intracellular capacity. The pathogen is resistant to penicillin, ampicillin first- and second-generation cephalosporins, gentamicin, tobramycin, and streptomycin. The intensive treatment phase involves intravenous administration of ceftazidime for 10-14 days or longer. This antibiotic has a bactericidal action that inhibits enzymes responsible for cell-wall synthesis in Gram- bacteria. A vaccine is under development but economic constraints may make vaccination an unrealistic option for many regions where Melioidosis is an issue.
References
Department of Agriculture, Center for Food Security and Public Health. 2003. Melioidosis. Ames (IA): Department of Agriculture, Center for Food Security and Public Health; [accessed 2017 Nov 11]. http://www.nj.gov/agriculture/divisions/ah/diseases/melioidosis.html
Adler NRL, Govan B, Cullinane M, Harper M, Adler B, Boyce JD. 2009. The molecular and cellular basis of pathogenesis in melioidosis: how does Burkholderia pseudomallei cause disease? FEMS Microbiology Reviews. [2017 Nov 11]; 33(6): 1079–1099. doi: 10.1111/j.1574-6976.2009.00189.x.
Cheng AC, Currie BJ. 2005. Melioidosis: Epidemiology, Pathophysiology, and Management. Clinical Microbiology Reviews. [2017 Nov 11]; 18(2): 383-416. doi: 10.1128/CMR.18.2.383-416.2005.
Burtnick MN, Brett PJ, Nair V, Warawa JM, Woods DE, Gherardini FC. 2008. Burkholderia pseudomallei Type III Secretion System Mutants Exhibit Delayed Vacuolar Escape Phenotypes in RAW 264.7 Murine Macrophages. Infection and Immunity. [2017 Nov 11]; 76(7): 2991-3000. doi: 10.1128/IAI.00263-08.
