By Kathryn Landry, Natalia Lorenc and Fiona Chan Pak Choon
Introduction
Cronobacter sakazakii is a rod-shaped Gram-negative opportunistic pathogen most commonly infecting infants and people with weakened immune systems. It was first isolated in 1980 by JJ Farmer III, who named the species Enterobacter sakazakii. In 2007, it was reclassified as C. sakazakii. This food-borne pathogen is capable of surviving in dry environments such as powdered infant formula.
Disease
C. sakazakii is primarily transmitted by the consumption of contaminated foods such as powdered infant formula (PIF), cereal, dried herbs, and pasta. PIF is the most frequent vector and contamination may be caused in the factories, when being prepared at home, or through improper storage. Contamination occurs in both hospitals and at home.
Although bacterial infection of this sort is unusual, it is quite harmful once it enters the body under the right conditions. When children are born, their immune systems are very weak, which makes it harder for them to fight pathogens. Infants affected by maternal disease, are premature, or had a traumatic delivery are more susceptible to infection. On the contrary, adults have a more developed immune system that is capable of eliminating the bacterium. If an infant survives an infection by C. sakazakii, delayed brain development and other neurological disorders are frequent. Additionally, infections often result in sepsis, meningitis, which is a brain inflammation, or necrotising enterocolitis, an intestinal infection. Symptoms of the disease include loss of appetite, fever, and irritability.
Epidemiology
Cronobacter sakazakii prevails in low-weight neonates and premature babies, whose immune systems are underdeveloped. It is also common in immunocompromised elderly, although individuals of all ages may be infected. This pathogen was first identified in England from an infected patient. In 1958, two neonates died from C. sakazakii induced meningitis. Since the first identification, cases have increased. In 1998, an outbreak in Belgium killed 12 neonates in intensive care.
Currently, C. sakazakii infects four to six infants in the United States every year. In 2013, the estimated mortality rate was 80%. The rarity of cases creates difficulties in determining these statistics.
Virulence
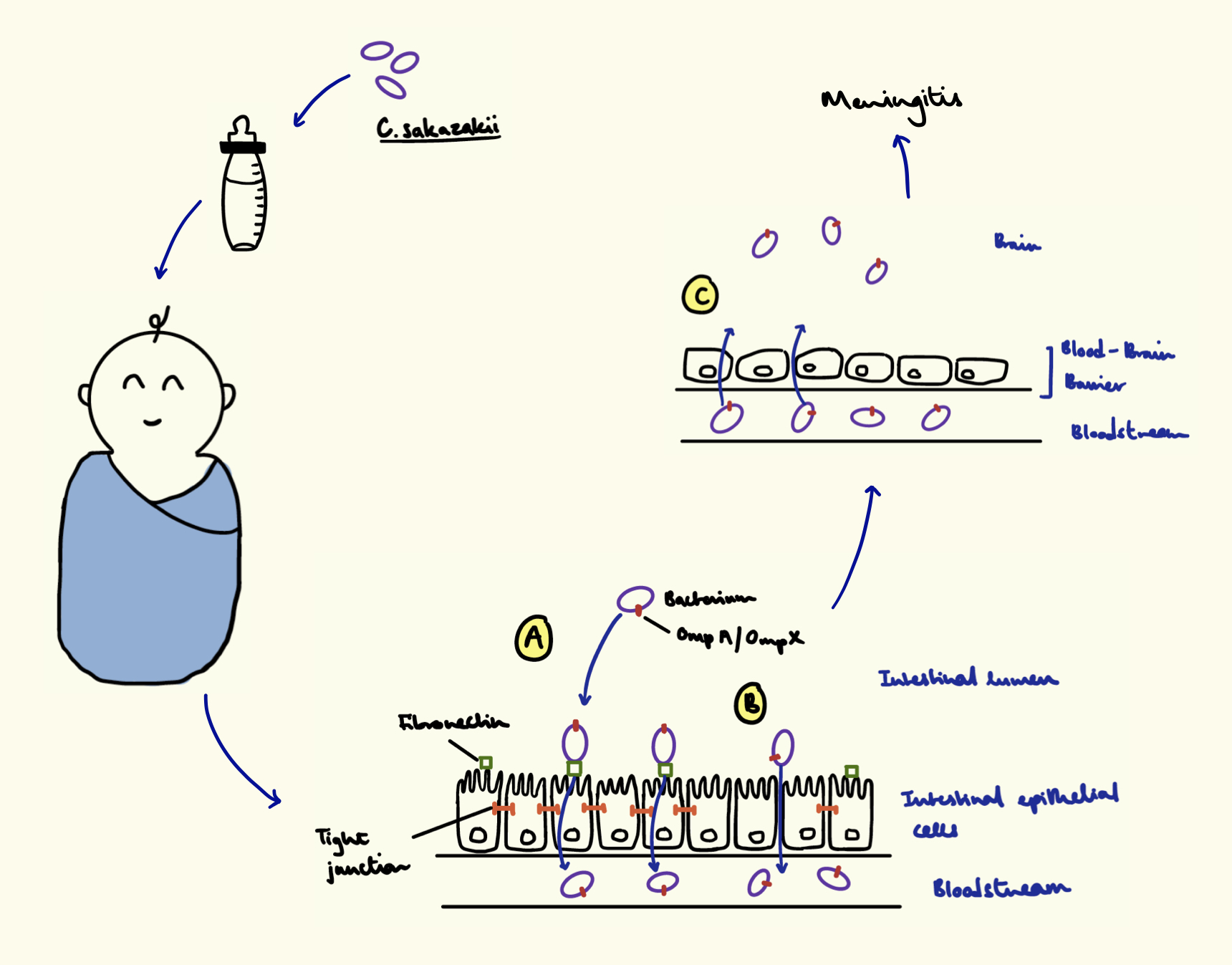
Since C. sakazakii is mainly a foodborne pathogen, it enters the host’s digestive tract via the mouth and then reaches its target site in the intestine. The bacterium adheres to the intestinal epithelial cells with the help of a glycoprotein called fibronectin (Figure 2A). In the host, fibronectin is found in the blood plasma and on cell surfaces. It helps mediate wound healing due to its ability to join 2 cells together by binding to both of these cells. By binding to the fibronectin present on the surface of intestinal epithelial cells, the pathogen adheres more strongly to the host tissue. The bacterium can also bypass the epithelium in newborns due to their underdeveloped tight junctions found between cells (Figure 2B). These junctions normally prevent such invaders to enter the bloodstream.
Once attached to its target cells, C. sakazakii invades the latter in order to enter the bloodstream, replicate, and proliferate. The bacterium contains 2 outer membrane proteins (Omp), OmpA and OmpX, which have an essential role in the invasion of intestinal cells (Figure 2A). After invading the intestinal cell, the pathogen moves across the cell before being released into the bloodstream via exocytosis – the transport of vesicles containing cellular components from the interior to the exterior of the cell. Once in the bloodstream, C. sakazakii travels to the blood-brain barrier before infecting the endothelial cells making up the barrier (Figure 2C). This causes meningitis, the inflammation of membranes surrounding the brain and the death of brain cells. OmpA and OmpX play a role in this secondary invasion, but their mechanism remains unknown.
C. sakazakii can also evade the host’s immune system by invading and replicating within macrophages – white blood cells that engulf and digest bacteria. It is believed that C. sakazakii possesses the ability to produce an enzyme called superoxide dismutase. This enzyme detoxifies potent compounds called reactive oxygen species (ROS) that are synthesized by macrophages to help digest bacteria. ROS can no longer degrade the pathogen and thus the pathogen survives and replicates. In addition, the pathogen is thought to produce enterotoxins that have yet to be determined.
Treatment
There are currently no available vaccines against C. sakazakii, but C. sakazakii is susceptible to several antibiotics including β-lactams, which inhibit the production of peptidoglycan, an important component of the bacterial cell wall. However, C. sakazakii contains several genes and mechanisms of antibiotic resistance, reducing the ability of the antibiotics to eliminate the infection. C. sakazakii has several efflux pumps, which remove the antibiotics from the cytoplasm and are therefore unable to locate their target and inhibit or kill the bacteria. In order to reduce the formation of antibiotic resistance, it is important to take the entire regimen of antibiotics the doctor prescribes.
References
Bowen AB and Braden CR. 2006. Invasive Enterobacter sakazakii Disease in Infants. Emerging Infectious Diseases. 12(8): 1185-1189.
Giri CP, Shima K, Tall BD, Curtis S, Sathyamoorthy V, Hanisch B, Kim KS, Kopecko DJ. 2012. Cronobacter spp. (previously Enterobacter sakazakii) invade and translocate across both cultured human intestinal epithelial cells and human brain microvascular endothelial cells. Microbial Pathogenesis 52:140–147.
Farmer JJ, Asbury MA, Hickman FW, Brenner DJ, and The Enterobacteriaceae Study Group. 1981. Enterobacter sakazakii: A new species of “Enterobacteriaceae” isolated from clinical specimens. International Journal of Systematic and Evolutionary Microbiology. 30(3): 583
Feeney A, Kropp KA, O’Connor R, Sleator RD. 2014. Cronobacter sakazakii: stress survival and virulence potential in an opportunistic foodborne pathogen. Gut Microbes 5:711–718.
Government of Canada [Internet]. 2012. [cited 2019 Nov 11]. Available from: https://www.canada.ca/en/public-health/services/food-poisoning/cronobacter.html
Henry M and Fouladkhah A. 2019. Outbreak History, Biofilm Formation, and Preventive Measures for Control of Cronobacter sakazakii in Infant Formula and Infant Care Settings. Microorganisms. 7(3): 1-10.
Hsiao C-T, Cheng H-W, Huang C-M, Li H-R, Ou M-H, Huang J-R, Khoo K-H, Yu HW, Chen Y-Q, Wang Y-K, et al. 2017. Fibronectin in cell adhesion and migration via N-glycosylation. Oncotarget 8:70653–70668.
Hunter CJ and Bean JF. 2013. Cronobacter: an emerging pathogen associated with neonatal meningitis, sepsis and necrotizing enterocolitis. Journal of Perinatology. 33: 581-585.
Joseph S and Forsythe SJ. 2011. Predominance of Cronobacter sakazakii Sequence Type 4 in Neonatal Infections. Emerging Infectious Diseases. 17(19): 1713-1715.
Kalyantanda G, Shumyak L, Archibald LK. 2015. Cronobacter Species Contamination of Powdered Infant Formula and the Implications of Neonatal Health. Frontiers in Pediatrics. 3: 56.
Manni ML, Tomai LP, Norris CA, Thomas LM, Kelley EE, Salter RD, Crapo JD, Chang L-YL, Watkins SC, Piganelli JD, et al. 2011. Extracellular Superoxide Dismutase in Macrophages Augments Bacterial Killing by Promoting Phagocytosis. The American Journal of Pathology 178:2752–2759.
McMullan R, Menon V, Beukers AG, Jensen SO, van Hal SJ, Davis R. 2018. Cronobacter sakazakii Infections from Expressed Breast Milk, Australia. Emerging Infectious Diseases. 24(2): 393-394.
