by Elizabeth Siciliani
Introduction
Shigella dysenteriae is a gram negative, rod-shaped (see figure 1), non-spore forming, facultative anaerobe (capable of both aerobic and anaerobic metabolism, depending on the availability of oxygen), nonmotile bacteria. As depicted in figure 1, this bacterium contains fimbriae, which are 1-2µm long, hair-like structures that allow efficient attachment to the host. The host cell and bacterial cell are both negatively charged in nature, which causes them to repel. Thus, the fimbriae contain adhesins (allow for stickiness) on their tips to contradict this repulsion. It is part of the Shigella genus, which can cause the acute intestinal illness, shigellosis. Shigellosis is a type of diarrhea caused by some bacteria of the Shigella genus, S. dysenteriae included. It is characterized by the phenotype of watery diarrhea at first, followed by the classic dysenteric stool, which is little in volume and grossly bloody. Shigellosis infects humans by invading the epithelial cells and multiplying within them, and eventually destroying them, causing the dysentery.
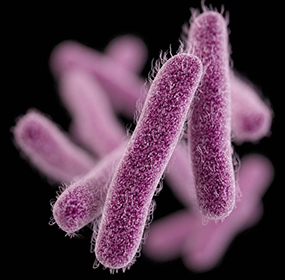
Figure 1: Computer-generated image of rod-shaped Shigella bacteria, based on electron micrograph images. The image depicts the fimbriae of Shigella. Source: Public Heath Image Library, Center for Disease Control (CDC).
Disease
Only humans and higher primates can carry Shigella spp.. No other species are affected by this pathogen, thus, it can only be transmitted via the fecal-oral route between humans. Transmission can occur through direct contact or sexually, as well as in contaminated food or water. They are a very infectious species, since as little as 10 microbes can cause disease.
The symptoms of Shigellosis include mucoid, bloody stools with detectable puss, along with abdominal cramps, fever, and tenesmus (the feeling of need to pass a stool despite an empty colon). In the intestinal epithelium, a strong inflammatory reaction will occur, which is responsible for ulcerations and abscess. This is due to both the killing of macrophages (cells of the immune system that are meant to uptake infectious agents and microorganisms) and disruption of the integrity of the intestinal epithelium as intestinal cells are killed.
Certain groups of individuals are at higher risk than others. Young children, travellers, men who have sex with men, and individuals with a compromised immune system.
Epidemiology
Worldwide, Shigella causes an estimated 80-165 million cases of disease per year, and 600,000 deaths.
The bacterium is endemic to temperate and tropical climates, and where hygiene is suboptimal. Compare to other species of Shigella, S. dysenteriae is less of a global health risk. It is most commonly isolated in sub-Saharan Africa and South Asia. Moreover, 56% of S. dyenteriae infections are associated with global travel. The countries that pose the highest risk for contraction of shigellosis are Africa, Central America, South America, and Asia in descending order.
Virulence factors
Shiga Toxin
Shiga toxin has been found to be an important virulence factor of S. dysenteriae. Its roles include inhibiting protein synthesis, inducing bloody diarrhea and causing hemorrhagic colitis and hemolytic uremic syndrome (HUS).
Shiga toxin is an AB5 toxin, which means that it is made up of one A subunit and five B subunits. The A subunit is responsible for conferring the enzymatic activity as it permanently inactivates the ribosome (a protein synthesis complex) of the host cell, and terminates all protein synthesis. The process begins when the B subunits bind to the host cell at a surface receptor, globotriaosylceramide (Gb3) (see figure 2). This initiates an uptake mechanism by the host cell, and eventually the toxin will gain access to the cytoplasm, which is the interior part of the host cell. When the toxin reaches this point, the A subunit can separate from the B5 subunit and elicit its function.

Figure 2: The trafficking of the AB5 shiga toxin. Drawing by: Elizabeth Siciliani. Adapted from: Valério et al. (2010).
Invasion of epithelial cells
There is a process that S. dysenteriae follows to successfully invade and infect the gastrointestinal epithelial cells (see figure 3). The first step is invasion of the M cells, or microfold cells, which are cells in the gastrointestinal epithelium, facing the lumen. They specialize in transporting antigen across to present to the underlying tissue. Antigen can be anything that the M cells “sample” from the intestinal lumen that does not come from our body. Normally, M cells would pass antigen to a macrophage, which is a professional antigen presenting cell (APC). Macrophages reside on the other side of the M cells (see figure 3). The problem arises at the level of the macrophage, when the M cell passes on the bacterium. Rather than the usual response of degradation in the macrophage, which would begin an immune mechanism, the intracellular pathogen specializes in hijacking the degradation pathway and not allowing it to happen. When the macrophage is exposed to shigella, it undergoes apoptosis (programmed cell death) and releases proinflammatory cytokine interleukin 1 (IL-1). Cytokines are small, soluble proteins that mediate inflammatory reactions and immunity.
This process begins with an S. dyenteriae protein, called IpaB protein; which both activates programmed cell death in macrophages, as well as turns pro-IL-1 into the active IL-1 form. By lysing the macrophage, the bacterium has access to the basolateral side of the epithelial cells (see figure 3). This is S. dysenteriae’s site of entry, where it penetrates the epithelial cell by forcing endocytosis. Shigella possesses a Type III secretion system (T3SS), which is a needle-like structure that penetrates the host cell membrane, and is an important virulence factor for cell entry. The T3SS injects a protein called IpaC into host cells, which causes the host cells to produce actin-rich filaments (a component of the cell’s skeleton). By doing this, the host cell is stimulated to produce arm-like structures called pseudopods that take up the bacterium, a process is called phagocytosis, which would not otherwise happen because epithelial cells are not phagocytic in nature. Once in the cell, the bacterium is in the correct location to multiply and eventually cause necrosis, which is an inflammatory, pathological form of cell death. S. dyenteriae can then spread laterally across the epithelium, spreading infection (see figure 3). They spread by causing an intracellular cascade of events in the host cell, that causes recruitment of the Arp2/3 complex. This complex recruits actin filaments, which make up the skeleton of the cell, defining its shape. The addition of actin is an important mechanism for the bacterium to move from cell to cell sideways, without ever going back out into the intestinal lumen. Staying inside the host cells is a mechanism of hiding from the humoral immune system, which protects from extracellular invaders and toxins. The addition of actin causes the walls of the infected host cell, along with the walls of the neighbouring host cell, to change shape to allow lateral spread of the bacterium (see figure 3). The wall of the cell extends out sideways, in a manner that allows the bacterium to push its way into the neighbouring cell. The strategy used for lateral spread is similar to the strategy used to get in the cell in the first place, where the bacteria manipulates the skeleton of the host cell so that it can initiate and spread infection.

Figure 3: Trafficking of Shigella dysenteriae through host cells. The process is described in the “Invasion of Epithelial Cells section of “Virulence Factors”. Drawing by: Elizabeth Siciliani. Adapted from: Hale TL, Keusch GT (1996).
Treatment
Shigellosis has the potential to resolve itself without treatment, if it did it would occur within 4-7 days. If shigellosis is acquired from international travel, antimicrobial treatment is administered; fluoroquinolone is of common use for shigellosis. However, it is prescribed with caution because of rising rates of multidrug resistance in Shigella spp., which is enhanced with overuse.
There is no existing vaccine unfortunately for Shigella, thus we cannot prevent or preclude disease, other than frequent hand washing, being mindful of cross-contamination in cooking and minimising fecal-oral contact during intercourse.
References
Donnenberg MS. Pathogenic strategies of enteric bacteria. Nature. 2000;406(6797):768-774. doi:10.1038/35021212.
Fontaine A, Arondel J, Sansonetti PJ. Role of Shiga toxin in the pathogenesis of bacillary dysentery, studied by using a Tox- mutant of Shigella dysenteriae 1. Infection and Immunity. 1988;56(12):3099-3109.
Gaston MA, Pellino CA, Weiss AA. Failure of Manganese to Protect from Shiga Toxin. PLoS ONE. 2013;8(7). doi:10.1371/journal.pone.0069823.
Gopal A, Chidambaram IS, Devaraj N, Devaraj H. Shigella dysenteriae infection activates proinflammatory response through β-catenin/NF-κB signaling pathway. Plos One. 2017;12(4). doi:10.1371/journal.pone.0174943.
Hale TL, Keusch GT. Shigella. 4th ed. (Baron S, ed.). Galveston, Texas: Medical Microbiology; 1996.
Keusch G.T., Acheson D.W.K. Shigella Infection. In: Paradise L.J., Bendinelli M., Friedman H. (eds) Enteric Infections and Immunity. Infectious Agents and Pathogenesis. Springer, Boston, MA; 1996.
Madigan MM. Brock Biology of Microorganisms. 14th ed. Harlow: Pearson Education Limited; 2014.
Shigella – Shigellosis. Centers for Disease Control and Prevention. https://www.cdc.gov/shigella/general-information.html. Published October 12, 2017. Accessed November 19, 2017.
Travelers’ Health. Centers for Disease Control and Prevention. https://wwwnc.cdc.gov/travel/yellowbook/2018/infectious-diseases-related-to-travel/shigellosis. Published May 31, 2017. Accessed November 19, 2017.
Valério E, Chaves S, Tenreiro R. Diversity and Impact of Prokaryotic Toxins on Aquatic Environments: A Review. Toxins. 2010;2(10):2359-2410. doi:10.3390/toxins2102359.
Wilson BA. Bacterial pathogenesis a molecular approach. 3rd ed. Washington, DC: ASM Press; 2011.
