by Christine L. Toma and Justine Hadrava
Introduction
Brucella abortus is an intracellular pathogen causing brucellosis, an infectious disease also referred to as undulant fever, Mediterranean fever or Malta fever. Brucellosis is categorized as a zoonosis, meaning that the infection primarily targets animals, but is also transmissible to humans. The principal hosts of B. abortus are livestock animals, but the clinical effects are more severe in humans. In addition to being of economic importance, B. abortus represents a threat to public health.
Disease
Predominantly in cattle, but also sometimes in sheep, goats and pigs, B. abortus infection is typically localized in the reproductive system of sexually mature animals. Both females and males can be affected, although more severe complications are observed in females. Brucellosis is usually characterized by abortion, premature birth, retained placenta and lowered milk production. Infertility occurs between two weeks to five months following the infection, but animals regain their reproductive capability afterwards. B. abortus transmission to humans is accomplished by direct or indirect contact with infected animals or animal products.
In humans, B. abortus induces severe illnesses involving fever, malaise, anorexia, depression, physical weakness, weight loss, headache and bone and muscle pain. It has the ability to spread to and damage any organ of the body and thus has the potential to cause what is called a systemic infection. In addition, all age groups are affected, contrary to animals. During pregnancy, B. abortus can cause spontaneous abortion, but can also be transmitted to the infant. The complications of the infection can be persistent and last for multiple weeks or months. Since the manifestations of B. abortus infection symptoms are highly variable and non-specific, laboratory tests need to be performed to confirm a diagnosis.
Epidemiology
Transmission between animals results mainly from contact following abortion or by ingestion of pasture contaminated with the bacteria. However, other inoculation source of B. abortus include infected milking devices and artificial insemination with contaminated semen. Susceptibility increases as the animal becomes sexually mature and decreases at least a thousand fold under vaccination. Transmission is also favored in high density herds. Animal brucellosis rarely leads to death, but the abortion rate of infected individuals is between 30% and 80%.
B.abortus is predominantly transmitted to humans from contaminated environments (Figure 1). Therefore, any farmworker, animal attendant, veterinarian, inseminator or worker involved in animal product processing is more susceptible to acquire brucellosis. B. abortus can survive for a long period of time in dung, water, dust, soil, meat and dairy products. Therefore, route of exposure includes inhalation of contaminated dust, dried dung etc., direct contact with an animal or its fluids, or accidental ingestion. For people who are not working with animals or animal parts, eating raw meat or unpasteurized milk or milk products is the major cause of B. abortus infection. The rate of brucellosis varies greatly between locations and can range from 0.01 to more than 200 cases per 100 000 inhabitants. However, in the US, brucellosis occurrence is less than 0.05 per 100 000 inhabitants. In addition, the mortality rate varies from 2% to 5% in untreated individuals and is extremely rare if appropriate treatment is received.
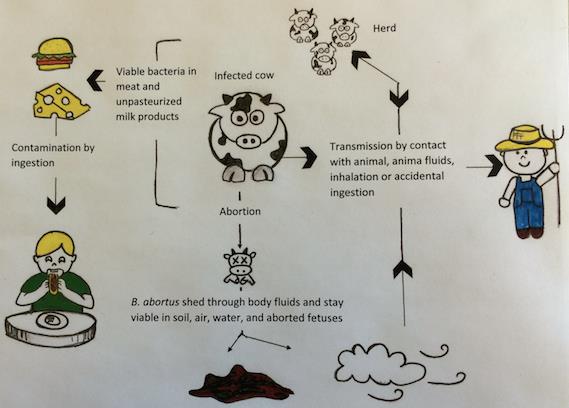
Figure 1: Summary of the major possible transmission routes of B. abortus to humans (figure by Justine Hadrava).
Virulence
Successful colonization of a host by B. abortus is achieved by molecular determinants that allow invasion, resistance to intracellular killing and replication in host cells. B. abortus invade the mucosa and are ingested by immune cells responsible for digesting bacteria called phagocytes. Specific molecules, such as a two component system and host cellular projections, are known to be responsible for the binding and integration of bacteria into host cells, but not all epithelial cells will allow amalgamation with B. abortus.
When the bacteria is ingested, it goes through a pathway to avoid phagolysosome fusion, a process in which pathogens are digested and killed by enzymes, and reach its replication niche (Figure 2). This bactericidal killing is not avoided by all bacteria, in fact after 48 hours only 15-30% of the immune cells display bacterial replication.
B. abortus possesses a particular lipopolysaccharide (LPS) on its outer membrane, a chemical structure that usually induces an immune response. Since this LPS is in a non-classical form, intracellular invasion and survival is more likely.
Replication occurs in membrane bound compartments resembling the endoplasmic reticulum (an organelle) of the cells (Figure 2). Bacteria can survive in cellular environments through the formation of autophagosome, a spherical structure formed by invagination of membrane structures surrounding B. abortus. A high rate of replication occurs when autophagosomes are fused with the endoplasmic reticulum, allowing bacteria to replicate and avoid the host’s immune system strategies. In pregnant animals, B. abortus replicates in the trophoblast, an outer layer that provides nutrients for the embryo and causes abortion.
Extensive replication of the bacteria occurs without disturbing basic cell functions. Necrosis is the final outcome of infected cells and when they burst, bacteria are released and the infection progresses (Figure 2).

Figure 2: B. abortus mechanism to avoid the host immune system within phagosomes (figure by Christine L. Toma).
Prevention and Treatment
Animals
The best way to deal with a disease is to prevent it. The key to prevent brucellosis in an animal herd is to be rigorous. It is important to always isolate newly purchased animals to make sure they are brucellosis free. Laboratory techniques used to identify the cause of abortion is also a way to single out sick animals. Good disposal of placentas and non-viable fetuses is important to reduce the risk of contamination. Hygiene and precautionary measures are the best allies for prevention.
The most effective way to control this disease is by vaccination. A strain of B. abortus is injected most commonly in sexually immature females to minimize the risk of abortion caused by the vaccine. It is known to be an effective method for elimination of clinical diseases and to reduce the number of organisms excreted per animal. Eradication of B. abortus is hard to achieve because it requires a surveillance system, laboratory support and a high level of hygiene.
Humans
To prevent human brucellosis, people should reduce exposure to infected animals or byproducts. Hygiene is the best preventative measure for workers who are in contact with animals on a daily basis; this applies to farm, meat production chain and laboratory personnel.
If infection is detected in an individual, an antibiotic treatment is usually prescribed for a specific duration of time. Sometimes a surgical intervention is needed, but full recovery is common.
References
Gorvel JP, Moreno E. 2002. Brucella intracellular life: from invasion to intracellular replication. Veterinary microbiology. 90: 281-297.
The Merk Veterinary Manual: Overview of Brucellosis in Cattle [Internet]. Date unknown. Kenilworth, N.J., U.S.A. Merck Sharp & Dohme Corp; [Updated 2013 July; cited 2015 November 10]. Available from: http://www.merckvetmanual.com/mvm/reproductive_system/brucellosis_in_large_animals/brucellosis_in_cattle.html
Corbel MJ, World Health Organization. 2006. Brucellosis in Humans and Animals.[Internet]. Albany, NY, USA: World Health Organization. [November 10 2015]. Available from: http://site.ebrary.com.proxy3.library.mcgill.ca/lib/mcgill/detail.action?docID=10190682
Corbel M.J. 2006. Brucellosis in human and animals. Geneva : World Health Organization.
The Center for food security and public health. 2009. Bovine brucellosis: brucella abortus.
