By: Aaron McCall, Audrey Roberge, Brett Moffit
Introduction:
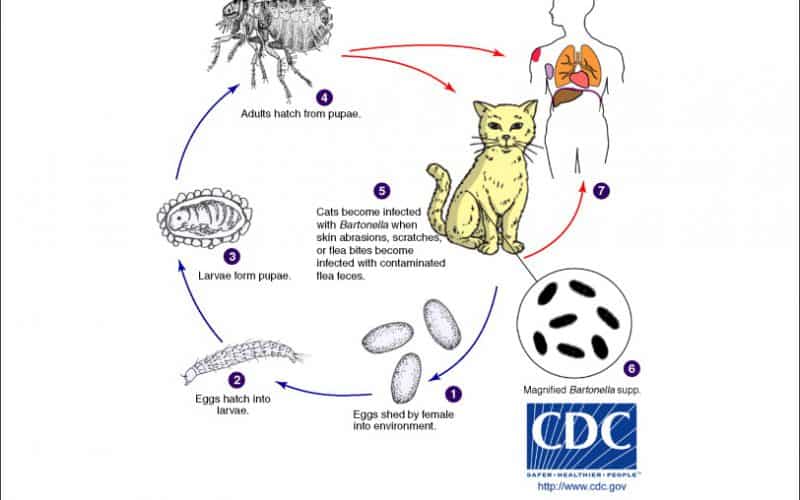
Bartonella henselae is the bacterial species that causes Bartonellosis, Cat Scratch Disease (CSD), or Cat Scratch Fever in humans. This species of Bartonella is a facultative intracellular Gram-negative bacteria that commonly infects cats and less commonly dogs. As it’s common name suggests, Cat Scratch Disease is contracted through the scratch or bite of an infected cat (Figure 1).
Disease:
The reservoir host for Bartonella henselae are cats which are therefore asymptomatic carriers. Cats contract B. henselae upon being infested with the common cat flea, Ctenocephalides felis where they reside in the gut and are expelled onto the cat’s skin via flea droppings. The cat becomes infected upon grooming itself which causes the bacteria contaminated flea feces to spread to its mouth and claws. Hence, transmission to humans results from the bite or scratch of an infected cat resulting in the inoculation of the flea feces and consequently the bacteria, into the open wound. Other potential vectors include additional blood sucking insects and arthropods such as ticks, lice and biting flies, namely the sand fly.
B. henselae causes disease primarily given the fact that it is able to evade the immune system by establishing an intracellular infection, as it is a facultative intracellular pathogen. Infection begins once the bacteria get inoculated through a bite or scratch wound and initially infect macrophages, the white blood cells that protect the body by “eating” pathogens. This allows the bacteria to subsequently infect the cells of the blood vessel lining, the endothelium. During this stage of infection, generally no symptoms are observed in immunocompetent individuals; however, bacillary angiomatosis is seen in immunocompromised patients. In this case, the bacteria trigger the endothelium lining cells to divide continuously, as well as block apoptosis, the ability for cells to self destruct. Therefore, resulting in the massive proliferation of the endothelium cells causing tumor-like structures that outgrow giving the appearance of papule lesions on the skin of the individual. Following infection of the endothelium, B. henselae can then infect red blood cells– also called erythrocytes– allowing them to establish a chronic infection as they are able to go undetected since red blood cells do not express MHC 1, an immune system activating signal protein that indicates that a cell has an intracellular pathogen. With the absence of MHC 1 the cytotoxic pathway of the adaptive immune system is not activated and therefore cannot effectively kill the intracellular pathogen. Infection in the blood also provides the bacteria with a highway by which it can travel to infect various organs. A common site of infection are the lymph nodes resulting in lymphadenopathy, which is severe regional inflammation of the lymph nodes. This is caused by the ongoing activation of the adaptive immune response as macrophages continually present B. henselae antigens to naive T cells,T cells who have not yet differentiated into a specific immunological role, either Killer T cells or Helper T cells. The continual presentation of B. henselae antigens to naive T cells triggers them to differentiate in order for the appropriate adaptive immune response to take place. However, since B. henselae establishes a proliferant intracellular infection of macrophages, this results in overwhelming the naive T cells to differentiate at a rate of which the lymphoid tissue can not keep up, resulting in hyperplasia of the lymph nodes, increasing the size of the tissue to compensate for the high demand of naive T cell differentiation. It has also been suggested that bartonella effector proteins (BEPs) also play a role in the presentation of this symptom, which will be discussed in greater detail in the virulence section. Another possible organ where infection takes place are the cells of the heart. This results in endocarditis and myocarditis– an inflammation of the heart– causing malfunctioning of the heart as it becomes more strenuous to pump blood throughout the body. This inflammation is clinically distinguished by the presence of heart murmurs.
Epidemiology:
The most common species of Bartonella found in infected cats is B. henselae. Within B. henselae, there are two different genotypes known to infect cats: Houston-1 (type 1), and Marseille (type 2). Type 1 B. henselae are more prevalent in Asia, while type 2 are found more commonly in western United States, western continental Europe, the United Kingdom, and Australia. Worldwide, infections of B. henselae are found more often in warm humid areas than in colder climates. However, regardless of the genotype that is most prevalent within a given country, the strains isolated from infected humans is most often type 1. The age and lifestyle of the cat also affects the probability of infection; young cats under the age of 1 year old and stray or feral cats are more likely bacteremic. This means that young, stray, or feral cats have a high probability of carrying or having this species of bacteria in their blood. Conversely, as cats get older, their probability of being seropositive for B. henselae increases. This is because as the young bacteremic cats age, their immune systems fight the infection and develop specialized antibodies designed to destroy the B. henselae cells present in the blood. After the cat recovers from the infection, the antibodies produced will persist in the blood and cause seropositivity. Antibody persistence is a part of acquired immunity and will help the cat fight off any subsequent infections of B. henselae more quickly and more efficiently.
Virulence Systems:
B. henselae employs specialized systems, called virulence factors, that help it colonize a host, infect the host’s cells, spread, and cause damage. The bacteria will infect the primary niches, which are the cells that help it migrate to the blood (macrophages and endothelial cells). The mechanism of this step is unclear, but the bacteria become competent after a priming period inside the primary niche, during which it stimulates inflammation but inhibits programmed cell death. It will then adhere to erythrocytes and force the erythrocyte to deform (with a protein called deformin) and internalize it, despite the erythrocyte’s usual lack of endocytic activity (internalizing something from outside the cell). Once in the erythrocyte, B. henselae can protect itself within a vacuole in the cell. This is also a potent virulence factor because it allows the bacteria to hide from the host’s immune defences. They also can evade the host’s immune system by hiding or modifying pathogen-associated molecular patterns which are patterns that the host would recognize as foreign bacteria. Once it has established itself in the blood, B. henselae will activate other virulence factors that are mostly involved in migrating from erythrocytes into other tissues. These mechanisms are poorly studied, although the bacteria express the same proteins that other types of bacteria employ, so these proteins likely are involved. When in other host cells, B. henselae use a type IV secretion system (T4SS) which delivers toxins directly to the host’s cells to release specific proteins called bartonella effector proteins (BEPs) that will cause the human host to produce a pathologic amount of erythrocytes so that the bacteria has more cells to colonize, as well as damaging the structure of many host cells to make them easier to invade. Other virulence factors cause hyperactivation of the adaptive immune system in lymph nodes (Figure 2), causing an intense immune response that will damage the host and direct tissue damage, especially to the heart, which is likely mediated by BEPs.

Treatment:
Generally, Bartonellosis subsides on its own with no medical intervention needed. A two-week treatment of Azithromycin, an antibiotic, is sometimes used to treat and diminish the time of lymphadenopathy presentation. A much lengthier treatment is required for more severe cases, especially in immunocompromised individuals, with an antibiotic treatment that can last up to six weeks. In this case, antibiotics of choice maybe be doxycycline, erythromycin and rifampin or any combination of the three medications. In cases where a patient has reached endocarditis, heart failure medications are also administered to manage the cardiac symptoms and valve replacement may be recommended.
References
Alexandre Harms, C. D. (2012). Intruders Below the Radar: Molecular Pathogenesis of Barontella spp. American Society for microbiology. Retrieved from: https://cmr.asm.org/content/25/1/42
Angelakis, E., & Raoult, D. (2014). Pathogenicity and treatment of Bartonella infections. International Journal of Antimicrobial Agents, 44(1), 16–25. doi: 10.1016/j.ijantimicag.2014.04.006
CDC. (n.d.). Bartonella Infection (Cat Scratch Disease, Trench Fever and Carrion’s Fever). Centers for Disease Control and Prevention. Retrieved from: https://www.cdc.gov/bartonella/index.html
Oskouizadeh, K., Zahraei-Salehi, T., & Aledavood, S. (2010). Detection of Bartonella henselae in domestic cats’ saliva. Iranian journal of microbiology, 2(2), 80–84. Sykes, J. E. (2014). Bartonellosis. In C. a. Diseases, Jane E Sykes (pp. 498-511). St Louis: Elsevier.
