By: Leen Bazzi & Stefanie Koch
Introduction: Gut Microbiome
Since the discovery of penicillin back in 1928, antibiotics have been used worldwide for many years to treat a wide array of bacterial infections. Extremely efficient with little to no short-term side effects in adults, antibiotics have been the first line of treatment against most bacterial infections. However, use of antibiotics has been strictly regulated due to the mounting evidence showing the influence of antibiotics on the gut microbiome and the function of the immune system.
Throughout our lifetime, as food passes through our gastrointestinal (GI) tract along with hundreds of microorganisms, a collection of microbes such as bacteria, archaea and eucaryotes, colonize our GI. They are called the ‘gut microbiota’ and they are normally not harmful to the host. In fact, they usually form a mutually beneficial relationship with the hosts, aiding in protecting against pathogens and even regulating host immune responses at times.
Recent research has shown that multiple factors such as dietary habits, stress and antibiotics can cause a microbiota dysbiosis, in other words, a change in the composition, diversity and function of the microbes in the gut. Antibiotics are now being recognized as a leading cause of gut dysbiosis in infants whose gut microbiota doesn’t fully develop and stabilize until the age of 3 years.
Gut dysbiosis in infants has been shown to affect their innate and adaptive immune systems through a reduction in the number of antibodies produced against a specific pathogen and changes in the phenotype and function of immune cells that attack foreign microorganisms, which reduces their ability to respond to signals and activate during infections in order to fight off the pathogen (Fig. 1).
A recent study by Aversa et al. published in 2021, in Mayo Clinic Proceedings, investigated the association between antibiotic exposure in the first two years of life and health conditions with childhood onset. They observed the medical records of 14,572 children, of whom 70% received at least one antibiotic prescription during the first two years of life and tracked their health records for any diseases developed between time of birth and 2017. Based on previous similar research, Aversa et al. hypothesized that intake of antibiotics during the first two years of life is correlated to an increased risk of immunological, metabolic, and neurobehavioral health conditions.
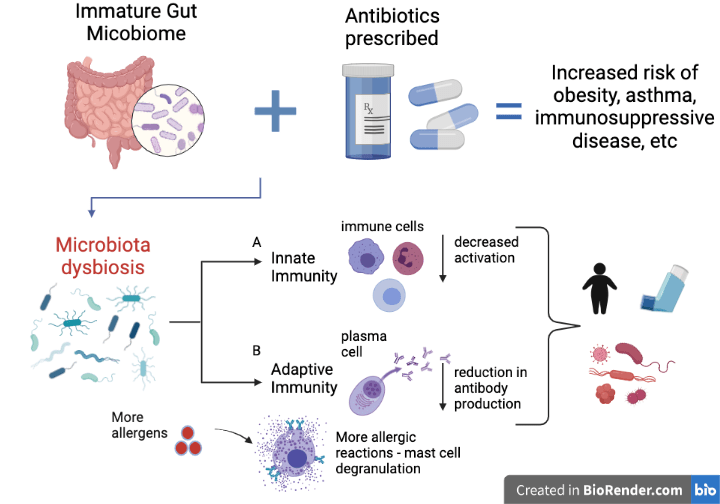
Fig.1 Summary of antibiotic-driven modulation of immune responses and possible consequences in infants under two years old. Antibiotic exposure causes microbiota dysbiosis. A. microbiota dysbiosis can change the phenotype and function of immune cells of the innate system leading to a decrease in activation. B. It can also cause a reduction in antibody production for certain antigens and mast cell degranulation. Source: Leen Bazzi, 2021 (BioRender)
Description of findings:
Overall, Aversa et al. (2021) found a significant association between exposure to certain antibiotics and increased probability of developing many health conditions. Specifically, obesity, asthma, allergic rhinitis, atopic dermatitis, ADHD and learning disabilities were among the most common health problems in kids exposed to two or more antibiotics.
The relationship between the number of antibiotics administered in infancy and the probability of developing 1,2 and 3 or more health conditions in those children was studied by Aversa et al.. They found that the probability of developing more than three health conditions in kids who have taken more than five antibiotics is significantly higher than in kids who have taken no antibiotics. Overall, kids exposed to more than one antibiotic had the highest probability of developing more than one health condition. This was observed with children that developed obesity and another condition such as asthma, ADHD, or a learning disability. However, obesity, asthma and allergic rhinitis was a common triad with exposed and non-exposed children. Atopic dermatitis or ADHD were more common to be paired with asthma and obesity in children exposed to antibiotics.
Aversa et al. also studied the correlation between the number of antibiotics administered and the time spent with 1, 2, 3 or more health conditions. On average, infants exposed to antibiotics spent more years with 2 or more health conditions. Infants exposed to more than 5 antibiotics had a significant increase in the years spent with more than one condition and a significant decrease in the years spent with no health conditions.
Furthermore, it seems the period of development in which the antibiotics were administered influences the risk of developing certain health problems. There was a much stronger risk of developing asthma, allergic rhinitis, and obesity when the children were given antibiotics between 6 and 12 months, a critical age for the development of gut microbiota.
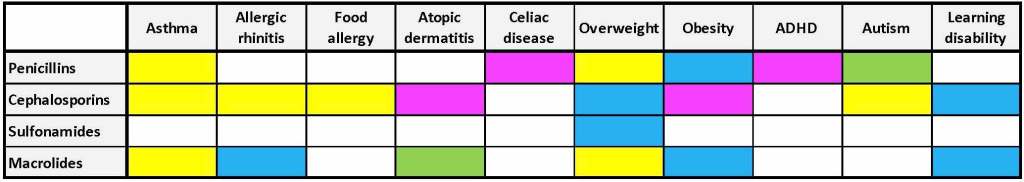
The health and neurobehavioral conditions developed varied based on the type of antibiotic exposed to and the genders of the patients. Many of them received penicillin (64%), while others received either Cephalosporins, Sulfonamides or Macrolides. There was a significant influence of antibiotic type on developing higher risk for certain conditions which also differed significantly between genders (Table 1). For example, when penicillin was prescribed, it had a higher risk to develop obesity in boys, and celiac disease in girls, while having a non-significant risk for food allergy in both genders. Obesity seems to be in large part more significant in boys for all 4 antibiotics. Autism and atopic dermatitis were less likely to occur in girls, if penicillin or macrolide were prescribed. Moreover, Cephalosporins seem to have the highest risk for development of autism in both sexes.
Table 1. Association between the type of antibiotic prescribed and the increased risk in developing health conditions. Highlighting the difference of conditions between genders, that were more significantly developed. The color assignments are shown in the legend and are as follows: pink for the significant results for girls; blue for boys; yellow for both genders; white the non-significant results for both genders; green for the significant decrease for girls. Source: Stefanie Koch, 2021. Data from Aversa et al. (2021)

Discussion:
The study tested the hypothesis that exposure to a certain number of antibiotics increases the risk of immunological and neurobehavioral diseases by causing microbiota dysbiosis, which could lead to disruption of the immune system in children (Fig. 1). The results of the study showed a significant correlation between the number of antibiotics taken before the age of two and an increase in the risk of developing certain diseases with childhood onset (Fig. 2). The association between antibiotic exposure and health outcomes in infants persisted even after the results were adjusted for important infant and maternal confounding variables such as, maternal age, delivery method, antibiotic exposure during pregnancy, etc. These variables could possibly skew the results, therefore, it’s important to look at a patients file closely before including them in the study. By doing so, the researchers reduced the variables that could skew the results, focusing more on the relation between the antibiotic and the related health outcomes. Thus, the findings support the hypothesis that antibiotics play a causal role in the pathogenesis of childhood immune disorders through disruption of microbiome during critical developmental periods.
The researchers also categorized certain health outcomes by the antibiotics the child was exposed to at a young age, as well as the different effects each antibiotic had on girls and boys. This allowed them to form a correlation between certain antibiotics and the risk for certain diseases based on sex (Table 1). Although the correlation found was not very significant, some antibiotics were associated with a higher risk of one or two diseases in a certain gender. These preliminary results could help with future studies about the different effects of antibiotics on females and males.
This population-based study is significant in supporting the hypothesis because of the large cohort of participants (14,572 children) and the long follow-up period ranging from 6-11 years for each participant. Additionally, the results of a significant association between antibiotics, gut health, and risks for developing health conditions is supported by other studies with similar results Also, choosing a suburban cohort for the study might have aided in reducing any exterior factors that could have affected the significance of the results, such as living conditions, environmental factors etc.
One limitation of the study is that the infections the children were treated for were not documented or taken into consideration. Maybe their illness may have had underlying effects or even directly led to the health condition seen later. Another limitation was the diversity in ethnicity of the study population, with 71% of the cohort being white, and only 29% encompassing all other groups. This might be because the study chose a population residing in Minnesota, USA. Therefore, the study can only apply to people living in the USA and not in other parts of the world where lifestyle, medical help and habits may differ.
Conclusion & Future Directives:
In conclusion, the study by Aversa et al. supports the hypothesis that there is a significant relationship between antibiotic exposure before the age of 2, when the microbes in the gut have not fully developed, and some diseases that develop later during childhood. The large cohort of 14,572 children shows that the results of this study are important and significant, even if they are only applicable in the USA. Furthermore, by analyzing how each antibiotic affected girls and boys, the study showed preliminary results indicating that antibiotics may have different effects depending on whether the patient is a female or male. The reason for this is unknown and more research is needed to study why the conditions developed differ between the sexes, regardless of the antibiotic prescribed. This could pave the way for different prescriptions based on the sex of the patient. Future studies about this topic should also emphasize diversity in ethnicity in order to see whether these results are significant in other ethnicities or whether they may differ based on lifestyle. Some research is currently being done about antibiotics that could strengthen the host immune system rather than simply attacking the bacteria causing the infection. This is something that could potentially overcome the issue of antibiotics changing the composition and function of gut microbiota if the study succeeds.
References
Aversa, Z., Atkinson, E. J., Schafer, M. J., Theiler, R. N., Rocca, W. A., Blaser, M. J., & LeBrasseur, N. K. (2021). Association of Infant Antibiotic Exposure With Childhood Health Outcomes. Mayo Clin Proc, 96(1), 66-77. doi:10.1016/j.mayocp.2020.07.019
Gensollen, T., Iyer, S. S., Kasper, D. L., & Blumberg, R. S. (2016). How colonization by microbiota in early life shapes the immune system. Science (New York, N.Y.), 352(6285), 539-544. doi:10.1126/science.aad9378
Gray, J., Oehrle, K., Worthen, G., Alenghat, T., Whitsett, J., & Deshmukh, H. (2017). Intestinal commensal bacteria mediate lung mucosal immunity and promote resistance of newborn mice to infection. Sci Transl Med, 9(376). doi:10.1126/scitranslmed.aaf9412
Hancock, R. E., Haney, E. F., & Gill, E. E. (2016). The immunology of host defence peptides: beyond antimicrobial activity. Nat Rev Immunol, 16(5), 321-334. doi:10.1038/nri.2016.29
Langdon, A., Crook, N., & Dantas, G. (2016). The effects of antibiotics on the microbiome throughout development and alternative approaches for therapeutic modulation. Genome Medicine, 8(1), 39. doi:10.1186/s13073-016-0294-z
Mitre, E., Susi, A., Kropp, L. E., Schwartz, D. J., Gorman, G. H., & Nylund, C. M. (2018). Association Between Use of Acid-Suppressive Medications and Antibiotics During Infancy and Allergic Diseases in Early Childhood. JAMA Pediatr, 172(6), e180315. doi:10.1001/jamapediatrics.2018.0315
Panda, S., El khader, I., Casellas, F., López Vivancos, J., García Cors, M., Santiago, A., . . . Manichanh, C. (2014). Short-Term Effect of Antibiotics on Human Gut Microbiota. PLOS ONE, 9(4), e95476. doi:10.1371/journal.pone.0095476
Roubaud-Baudron, C., Ruiz, V. E., Swan, A. M., Jr., Vallance, B. A., Ozkul, C., Pei, Z., . . . Blaser, M. J. (2019). Long-Term Effects of Early-Life Antibiotic Exposure on Resistance to Subsequent Bacterial Infection. mBio, 10(6). doi:10.1128/mBio.02820-19
Shekhar, S., & Petersen, F. C. (2020). The Dark Side of Antibiotics: Adverse Effects on the Infant Immune Defense Against Infection. Frontiers in Pediatrics, 8(651). doi:10.3389/fped.2020.544460
Thursby, E., & Juge, N. (2017). Introduction to the human gut microbiota. Biochemical Journal, 474(11), 1823-1836. doi:10.1042/bcj20160510
Zhu, D., Xiao, S., Yu, J., Ai, Q., He, Y., Cheng, C., . . . Pan, Y. (2017). Effects of One-Week Empirical Antibiotic Therapy on the Early Development of Gut Microbiota and Metabolites in Preterm Infants. Sci Rep, 7(1), 8025. doi:10.1038/s41598-017-08530-9
